2013-14 Departmental Performance Report
Table of Contents
- Foreword
- Minister's Message
- Section I: Organizational Expenditure Overview
- Section II: Analysis of Programs by Strategic Outcome
- Strategic Outcome: A world-class health research enterprise that creates, disseminates and applies new knowledge across all areas of health research.
- Program 1.1: Health Knowledge
- Program 1.2: Health Researchers
- Program 1.3: Health Research Commercialization
- Program 1.4: Health and Health Services Advances
- Sub-Program 1.4.1: Institute Strategic Initiatives
- Sub-Sub-Program 1.4.1.1: Strategic Initiatives Program
- Sub-Sub-Program 1.4.1.2: Institute Support Grants Program
- Sub-Sub-Program 1.4.1.3: HIV/AIDS Research Initiative
- Sub-Sub-Program 1.4.1.4: Pandemic Preparedness Strategic Research Initiative
- Sub-Sub-Program 1.4.1.5: National Anti-Drug Strategy Treatment Research Initiative
- Sub-Sub-Program 1.4.1.6: Strategy for Patient-Oriented Research Initiative
- Sub-Sub-Program 1.4.1.7: Drug Safety and Effectiveness Network Initiative
- Sub-Program 1.4.2: Knowledge Translation Programs
- Sub-Program 1.4.1: Institute Strategic Initiatives
- Internal Services
- Strategic Outcome: A world-class health research enterprise that creates, disseminates and applies new knowledge across all areas of health research.
- Section III: Supplementary Information
- Section IV: Organizational Contact Information
- Appendix: Definitions
- Endnotes
Foreword
Departmental Performance Reports are part of the Estimates family of documents. Estimates documents support appropriation acts, which specify the amounts and broad purposes for which funds can be spent by the government. The Estimates document family has three parts.
Part I (Government Expenditure Plan) provides an overview of federal spending.
Part II (Main Estimates) lists the financial resources required by individual departments, agencies and Crown corporations for the upcoming fiscal year.
Part III (Departmental Expenditure Plans) consists of two documents. Reports on Plans and Priorities (RPPs) are expenditure plans for each appropriated department and agency (excluding Crown corporations). They describe departmental priorities, strategic outcomes, programs, expected results and associated resource requirements, covering a three-year period beginning with the year indicated in the title of the report. Departmental Performance Reports (DPRs) are individual department and agency accounts of actual performance, for the most recently completed fiscal year, against the plans, priorities and expected results set out in their respective RPPs. DPRs inform parliamentarians and Canadians of the results achieved by government organizations for Canadians.
Additionally, Supplementary Estimates documents present information on spending requirements that were either not sufficiently developed in time for inclusion in the Main Estimates or were subsequently refined to account for developments in particular programs and services.
The financial information in DPRs is drawn directly from authorities presented in the Main Estimates and the planned spending information in RPPs. The financial information in DPRs is also consistent with information in the Public Accounts of Canada. The Public Accounts of Canada include the Government of Canada Consolidated Statement of Financial Position, the Consolidated Statement of Operations and Accumulated Deficit, the Consolidated Statement of Change in Net Debt, and the Consolidated Statement of Cash Flow, as well as details of financial operations segregated by ministerial portfolio for a given fiscal year. For the DPR, two types of financial information are drawn from the Public Accounts of Canada: authorities available for use by an appropriated organization for the fiscal year, and authorities used for that same fiscal year. The latter corresponds to actual spending as presented in the DPR.
The Treasury Board Policy on Management, Resources and Results Structures further strengthens the alignment of the performance information presented in DPRs, other Estimates documents and the Public Accounts of Canada. The policy establishes the Program Alignment Architecture of appropriated organizations as the structure against which financial and non-financial performance information is provided for Estimates and parliamentary reporting. The same reporting structure applies irrespective of whether the organization is reporting in the Main Estimates, the RPP, the DPR or the Public Accounts of Canada.
A number of changes have been made to DPRs for 2013−14 to better support decisions on appropriations. Where applicable, DPRs now provide financial, human resources and performance information in Section II at the lowest level of the organization's Program Alignment Architecture.
In addition, the DPR's format and terminology have been revised to provide greater clarity, consistency and a strengthened emphasis on Estimates and Public Accounts information. As well, departmental reporting on the Federal Sustainable Development Strategy has been consolidated into a new supplementary information table posted on departmental websites. This new table brings together all of the components of the Departmental Sustainable Development Strategy formerly presented in DPRs and on departmental websites, including reporting on the Greening of Government Operations and Strategic Environmental Assessments. Section III of the report provides a link to the new table on the organization's website. Finally, definitions of terminology are now provided in an appendix.
Minister's Message

I am pleased to present the 2013-14 Canadian Institutes of Health Research's (CIHR) Departmental Performance Report.
Knowledge gained through health research helps improve our health care system and generate new opportunities for health innovation. Through CIHR, the Government of Canada continues to invest in health research excellence and innovation.
Since releasing its five-year strategic plan, Health Research Roadmap, in 2009, CIHR has been working towards a specific set of objectives for investing in world-class research, addressing health and health-system priorities, and translating research findings into benefits for Canadians. Over the past year, the organization has been developing a refresh of its strategic plan, in order to build on the work completed to date and ensure alignment with the health research priorities of Canadians.
CIHR continued to roll out the reforms of its Open Suite of Programs and peer review process. Milestones achieved this year include the completion of the first two pilot studies and the launch of the first Foundation Scheme pilot competition. The pilots were developed to test new features which have contributed to refinements of the overall design. The reforms are aimed at ensuring the long-term sustainability of a robust health research environment in Canada, and they will continue over the next several years.
CIHR also continued to invest in priority-driven research through such initiatives as the Strategy for Patient-Oriented Research (SPOR). This year saw the launch of the first SUPPORT (Support for People and Patient-Oriented Research and Trials) units in Alberta and Manitoba. CIHR also launched a call for expressions of interest for the SPOR Network in Youth and Adolescent Mental Health. The SPOR SUPPORT units and the SPOR networks will help address high priority health challenges and bridge the gap between research evidence and health care practice.
Through its work, CIHR invests, builds partnerships and helps catalyze research initiatives that are generating important health innovations. As it moves forward with its new strategic plan, CIHR will continue to play a key role in improving the health of Canadians.
The Honourable Rona Ambrose, P.C., M.P.
Minister of Health
Section I: Organizational Expenditure Overview
Organizational Profile
- Appropriate Minister: Rona Ambrose
- Institutional Head: Dr. Alain Beaudet
- Ministerial Portfolio: Health
- Enabling Instrument(s): Canadian Institutes of Health Research Act (S.C. 2000, c. 6)Footnote 1
- Year of Incorporation / Commencement: 2000
Organizational Context
Raison d'être
The Canadian Institutes of Health ResearchFootnote 2 (CIHR) is the Government of Canada's health research funding agency. It was created with a mandate "to excel, according to internationally accepted standards of scientific excellence, in the creation of new knowledge and its translation into improved health for Canadians, more effective health services and products and a strengthened Canadian health care system."
Responsibilities
CIHR was designed to respond to the evolving needs for health research and seeks to transform health research in Canada by:
- funding both investigator-initiated research and research on targeted priority areas;
- building research capacity in underdeveloped areas and training the next generation of health researchers; and
- focusing on knowledge translation that facilitates the application of research results and their transformation into new policies, practices, procedures, products and services.
CIHR integrates research through a unique interdisciplinary structure made up of 13 "virtual" institutes.Footnote 3 These institutes are not "bricks and mortar" buildings but communities of experts in specific areas. Collectively, the institutes support a broad spectrum of research: biomedical; clinical; health systems and services; and the social, cultural and environmental factors that affect the health of populations. Institutes form national research networks linking researchers, funders and knowledge users across Canada to work on priority areas.
CIHR reports through the Minister of HealthFootnote 4 and plays a key role in the Health Portfolio. As Canada's health research funding agency, CIHR makes an essential contribution to the Minister of Health's overall responsibilities by funding the research and knowledge translation needed to inform the evolution of Canadian health policy and regulation, and by taking an advisory role on research and innovation issues. This is achieved through an extensive and growing set of linkages with Health Canada and the Public Health Agency of Canada, providing decision makers with access to high-quality and timely health research.
CIHR works closely with the Natural Sciences and Engineering Research CouncilFootnote 5 (NSERC) and the Social Sciences and Humanities Research CouncilFootnote 6 (SSHRC), the two granting councils of the Industry PortfolioFootnote 7, to share information and coordinate efforts, harmonize practices, avoid duplication and foster multidisciplinary research. The three organizations (referred to as "Tri-Agency") provide a channel for the implementation of common policies, practices and approaches, whenever possible.
CIHR's Governing CouncilFootnote 8 (GC) sets the strategic direction of the agency and is responsible for evaluating its performance. Leadership on research, knowledge translation and funding for research is provided by the Science CouncilFootnote 9 (SC), while leadership on corporate policy and management is provided by the Executive Management CommitteeFootnote 10 (EMC).
Strategic Outcome and Program Alignment Architecture
CIHR's Program Alignment Architecture (PAA), was approved by the Treasury Board of Canada Secretariat in May 2009 and consists of one Strategic Outcome and five Programs that support the Strategic Outcome.
In 2013-14, CIHR developed a new PAA structure which was officially implemented in April 2014 for the 2014-15 fiscal year. This new structure will ensure that programs and sub-programs are clear, allow for proper alignment and reporting, and reduce the amount of overlap between different levels of the PAA that were present previously.
The performance information presented in Section II is organized according to the 2013-14 PAA structure as shown below:
- 1. Strategic Outcome: A world-class health research enterprise that creates, disseminates and applies new knowledge across all areas of health research
- 1.1 Program: Health Knowledge
- 1.1.1 Sub-Program: Open Research Grant Program
- 1.1.2 Sub-Program: Randomized Controlled Trials Program
- 1.2 Program: Health Researchers
- 1.2.1 Sub-Program: Salary Support Programs
- 1.2.1.1 Sub-Sub-Program: Open Salary Support Programs
- 1.2.1.2 Sub-Sub-Program: Canada Research Chairs Program
- 1.2.1.3 Sub-Sub-Program: Canada Excellence Research Chairs Program
- 1.2.2 Sub-Program: Training Support Programs
- 1.2.2.1 Sub-Sub-Program: Open Training Support
- 1.2.2.2 Sub-Sub-Program: Sir Frederick Banting and Dr. Charles Best Canada Graduate Scholarships Program
- 1.2.2.3 Sub-Sub-Program: Georges Philias Vanier Canada Graduate Scholarships Program
- 1.2.1 Sub-Program: Salary Support Programs
- 1.3 Program: Health Research Commercialization
- 1.3.1 Sub-Program: Research Commercialization Program
- 1.3.2 Sub-Program: Networks of Centres of Excellence Programs
- 1.3.2.1 Sub-Sub-Program: Networks of Centres of Excellence Program
- 1.3.2.2 Sub-Sub-Program: Business-Led Networks of Centres of Excellence Program
- 1.3.2.3 Sub-Sub-Program: Centres of Excellence for Commercialization and Research Program
- 1.4 Program: Health and Health Services Advances
- 1.4.1 Sub-Program: Institute Strategic Initiatives
- 1.4.1.1 Sub-Sub-Program: Strategic Initiatives Program
- 1.4.1.2 Sub-Sub-Program: Institute Support Grants Program
- 1.4.1.3 Sub-Sub-Program: HIV/AIDS Research Initiative
- 1.4.1.4 Sub-Sub-Program: Pandemic Preparedness Strategic Research Initiative
- 1.4.1.5 Sub-Sub-Program: National Anti-Drug Strategy Treatment Research Initiative
- 1.4.1.6 Sub-Sub-Program: Strategy for Patient-Oriented Research Initiative
- 1.4.1.7 Sub-Sub-Program: Drug Safety and Effectiveness Network Initiative
- 1.4.2 Sub-Program: Knowledge Translation Programs
- 1.4.1 Sub-Program: Institute Strategic Initiatives
Internal Services
- 1.1 Program: Health Knowledge
Organizational Priorities
In 2009, CIHR's Governing Council (GC) approved CIHR's second strategic plan (2009-14), Health Research Roadmap: Creating innovative research for better health and health care.Footnote 11 This strategic plan is the product of widespread consultations with members of the health research community, careful assessment of Canada's strengths and weaknesses, and ongoing deliberation about what CIHR would like to achieve by 2014. Roadmap sets out a vision comprising four strategic directions that are aligned with CIHR's corporate, business and operational priorities.
In 2013-14, CIHR continued to implement Roadmap while beginning work on a renewed strategic plan, which presents CIHR's continued vision based on its achievements to date, as well as the desire to capture excellence and accelerate health innovation to improve the health of Canadians. CIHR has engaged partners and stakeholders in order to deliver a national vision for health research in Canada and launch Roadmap II in 2014-15.
| Priority | Type | Programs |
|---|---|---|
| Invest in world-class research excellence | Ongoing | 1.1 Health Knowledge 1.2 Health Researchers |
Summary of ProgressCIHR continued to invest in the best ideas and brightest minds and enabled students to gain research experience by funding 798 new grants in 2013-14 through the Open Operating Grant ProgramFootnote 12 competitions. Approximately 25% of grant budgets were used to fund trainees. CIHR is also attracting and retaining the best researchers through a number of Tri-AgencyFootnote 13 mechanisms such as the Canada Graduate ScholarshipsFootnote 14, the Vanier Canada Graduate ScholarshipsFootnote 15 and the Banting Postdoctoral FellowshipsFootnote 16, as well as the Canada Research ChairsFootnote 17 and the Canada Excellence Research Chairs.Footnote 18 Overall in 2013-14, CIHR supported 1,472 recipients through these programs in order to attract the world's top researchers and graduate students, and promote the development and application of leading-edge knowledge. Other initiatives such as Science to BusinessFootnote 19, Science Policy FellowshipsFootnote 20 and the Strategic Training Initiative in Health ResearchFootnote 21 help break professional and sectoral barriers in health research and promote the development of transferable skills. These programs represent an ongoing investment of $15.1M in 2013-14 for 79 grants and awards. In 2013-14, CIHR began implementing its reforms of the Open Suite of Programs and peer reviewFootnote 22 processes. Two pilots were completed this year to test elements of the design. The results of the Fellowship pilotFootnote 23 are available on the CIHR website.Footnote 24 The first Knowledge Synthesis pilot was conducted, and a report will be published in 2014-15. A number of key components were developed to support the first external deployment of the College of ReviewersFootnote 25 in 2014, most notably, the recruitment of an external advisory committee. The committee has helped CIHR refine the design of the College to support the first deployment, including the process for validating, recruiting and enrolling reviewers, and the terms and conditions for joining the College of Reviewers. |
||
| Priority | Type | Program |
|---|---|---|
| Address health and health system research priorities | Ongoing | 1.4 Health and Health Services Advances |
Summary of ProgressCIHR invested in priority areas and addressed gaps in key research areas and communities, through development and implementation of eight signature initiatives.Footnote 26 The signature initiatives are in various phases of development, but progress over the last year includes the launch of funding opportunities, submission of letters of intent and grant applications, funding of successful applications, and end of grant reporting. An example for one initiative is identifying the successful teams in the letter of intent phase for the "European Research Projects on Rare Diseases"Footnote 27 ERA-Rare competition, and inviting them to apply with full applications to the competition. The Strategy for Patient-Oriented ResearchFootnote 28 (SPOR), a major priority for CIHR and the Government of Canada, made significant progress this year. In 2013-14, business plans for six SUPPORT unitsFootnote 29 were received, reviewed and approved for funding; grant agreements for SUPPORT units in Alberta ($24.3M), Manitoba ($22.4M), Ontario ($52.1M) and the Maritimes ($8.8M), which include Prince Edward Island, New Brunswick and Nova Scotia, were negotiated and signed. Two such agreements were announced by the federal Minister of Health and her provincial counterparts in Alberta and Manitoba. SUPPORT unit representatives are working together to implement this foundational element of SPOR. |
||
| Priority | Type | Programs |
|---|---|---|
| Accelerate the capture of health and economic benefits of health research | Ongoing | 1.3 Health Research Commercialization 1.4 Health and Health Services Advances |
Summary of ProgressCIHR developed strategic partnerships with key stakeholders including industry, provincial governments and charities to fund collaborative research that will benefit Canadians. For example, the pan-Canadian research networks in respiratory health, stroke, and vascular health are leveraging $9.7M over five years in additional funding from multiple partners. The grant agreements for SUPPORT units in Alberta ($24.3M), Manitoba ($22.4M), Ontario ($52.1M) and the Maritimes ($8.8M) (which include Prince Edward Island, New Brunswick and Nova Scotia) have resulted in a five-year investment totalling $107.6M to date, split equally between CIHR and the provinces. A new commercialization strategy is in the early stages of development at CIHR. Stakeholder consultations were completed in 2013-14 and will help guide the development of the strategy. An evaluation of the CIHR commercialization programs began in 2013-14 and will be completed in 2014-15. The results of the evaluation will contribute to the design and implementation of the strategy. CIHR, in collaboration with Canada's Research Based Pharmaceutical CompaniesFootnote 30 (Rx&D) and HealthCareCANFootnote 31 (formally the Association of Canadian Academic Healthcare Organizations and the Canadian Healthcare Association) implemented the Canadian Clinical Trials Coordinating Centre.Footnote 32 This investment of $1.5M over three years was created to help reverse the decline in clinical trial investment in Canada and improve the clinical trial landscape. CIHR is also committed to ensuring that knowledge creation is harnessed to drive health innovation forward. In December 2013, Canadian experts in dementia research joined counterparts from other G8 countries to discuss the current state of knowledge about dementia and define future international collaborative research efforts. The countries agreed to work together to double funding for dementia research and increase the number of people involved in clinical trials. Canada, with France as its partner, will host one of four follow-up legacy workshops in 2014-15. |
||
| Priority | Type | Program |
|---|---|---|
| Achieve organizational excellence, foster ethics and demonstrate impact | Ongoing | Internal Services |
Summary of ProgressIn 2013-14, CIHR undertook a comprehensive review of its multi-year strategic investment plan process to ensure that sound financial, performance measurement and risk management practices are considered at all stages of development and approval. As a result, CIHR's multi-year strategic investment plan was revised to include clear accountabilities, timely financial and performance measurement reporting and sufficient operating resources. In 2013-14, CIHR reached a new milestone with the introduction of pilot competitions for the new Open Suite of Programs and peer review processes. The pilots held in 2013Footnote 33 tested specific elements of the new competition processes (e.g. structured application, remote structured review). Through surveys to applicants and reviewers, CIHR identified what is working well and what needs refinement. These lessons are being incorporated into future pilots and into the final design of the new Open Suite of Programs and peer review processes. CIHR was successful not only in engaging multiple partners financially, but also in ensuring that appropriate partners, stakeholders and patients were engaged in the implementation of SPOR and other CIHR signature initiatives. As part of SPOR, a variety of stakeholders were invited to participate in CIHR-led workshops and meetings around all elements of the initiative, thus ensuring their concerns and expertise are considered in its further development and implementation. The creation of a framework on capacity development in patient-oriented research is in progress and is being informed through a number of sources including recommendationsFootnote 34 from an external advisory committee commissioned by the SPOR National Steering CommitteeFootnote 35 to provide guidance on the development of a training and career development strategy, as well as feedback from a March 2014 workshop on capacity development with relevant SPOR stakeholders. |
||
Risk Analysis
From its inception, CIHR has looked to establish effective partnerships with researchers, other federal departments and agencies, other national governments, non-government organizations, not-for-profit organizations and the private sector, to identify and address the health needs of Canadians and invest in health research innovation. Through collaboration with its network of partners and key stakeholders, CIHR has been able to leverage funding to increase its investment impact in health research over the past three years and ensure better mobilization, translation and diffusion of newly discovered knowledge and research resulting from the academic and private sectors. As a result, CIHR is making a difference in the lives of Canadians.
In the 2013 Speech from the ThroneFootnote 36, the Government of Canada committed to releasing an updated Science, Technology and Innovation Strategy, as well as to continuing support for science and innovation. This is an opportunity for CIHR to further support health research innovation.
CIHR continues to adapt to its ever-changing environment in an effort to ensure that the health research it funds contributes to the health and well-being of Canadians. Given this new context, CIHR began to refresh its five-year strategic plan in 2013-14 and will seek to increase the number of partners at all levels in priority research areas and to continue to leverage funds from a variety of sources, both public and private, to ensure that it is able to deliver on its key commitments.
As part of the renewal of its strategic plan, CIHR developed a new Performance Measurement Regime that will inform decision making at CIHR and allow for improved reporting both internally and externally. CIHR significantly updated its Corporate Risk Profile for 2014-15.
In 2013-14, CIHR identified 13 risks in its Corporate Risk Profile, of which four were considered high risks requiring ongoing mitigation and monitoring. These four risks are outlined below.
2013-14 Key Risks
| Risk 1 - Reform of the Open Suite of Programs | Link to Program Alignment Architecture | Link to Organizational Priorities |
|---|---|---|
| In 2013-14, a risk was identified that CIHR may be unable to fully deliver on the reforms being made to the Open Suite of Programs, including changes to the peer review process. These changes are pivotal for the implementation of Health Research Roadmap. This includes the risk that external stakeholders do not understand or support the proposed changes to the programs and/or peer review processes. | Strategic Outcome: A world-class health research enterprise that creates, disseminates and applies new knowledge across all areas of health research | Invest in World-Class Research Excellence |
Risk Response Strategy:The Reforms of the Open Suite of ProgramsFootnote 37 was identified in the 2013-14 Report on Plans and Priorities (RPP) and 2013-14 Corporate Risk Profile (CRP). CIHR responded to this risk by:
Additionally, the following activities which were not included in the 2013-14 RPP were completed:
While much was accomplished to mitigate this risk, the Reforms of the Open Suite of Programs continues to be high risk and will be actively managed by CIHR in future. |
||
| Risk 2 - Knowledge Translation | Link to Program Alignment Architecture | Link to Organizational Priorities |
|---|---|---|
| Given CIHR's lack of direct control over factors influencing the uptake and use of the research it funds, in 2013-14 a risk was identified that CIHR may not be able to fully achieve the knowledge translation (KT) component of its mandate and improve the health of Canadians through health research. | Program 1.3 Health Research Commercialization Program 1.4 Health and Health Services Advances |
Accelerate the Capture of Health and Economic Benefits of Health Research |
Risk Response Strategy:The Knowledge Translation risk was identified in the 2013-14 RPP and 2013-14 CRP. CIHR responded to this risk by:
Additionally, CIHR performed the following mitigation strategies not identified in the 2013-14 RPP:
Knowledge Translation is now a moderate risk for CIHR and, as a result of its mitigation strategies and the implementation of key components; it has been removed from the Corporate Risk Profile for the 2014-15 fiscal year. |
||
| Risk 3 - Results Management and Monitoring | Link to Program Alignment Architecture | Link to Organizational Priorities |
|---|---|---|
| Performance reporting and evaluation are time-consuming, costly and at times burdensome to target audiences. In 2013-14, a risk was identified that CIHR may be unable to optimally and efficiently evaluate and report on its performance as well as on the results of funded research, which would compromise our ability to be accountable to Canadians. | Internal Services | Achieve organizational Excellence, Foster Ethics and Demonstrate Impact |
Risk Response Strategy:The Results Management and Monitoring risk was identified in the 2013-14 RPP and 2013-14 CRP. CIHR responded to this risk by:
Additionally, the following activities which were not included in the 2013-14 RPP were completed:
Although Results Management and Monitoring remained a high risk for CIHR during the 2013-14 fiscal year, it has been reduced to medium for the 2014-15 fiscal year. |
||
| Risk 4 - Institute Review | Link to Program Alignment Architecture | Link to Organizational Priorities |
|---|---|---|
| Due to the institute virtual organizational model, in 2013-14 CIHR identified a risk of disruptions and corporate memory loss during the transition period from an outgoing Scientific Director (SD) to an incoming. This may compromise the Institutes' abilities to achieve planned outcomes or their mandate in support of CIHR's strategic objectives. | Program 1.4 Health and Health Services Advances Internal Services | Address Health and Health System Research Priorities Achieve Organizational Excellence, Foster Ethics and Demonstrate Impact |
Risk Response Strategy:The Institute Review risk was identified in the 2013-14 RPP and 2013-14 CRP. CIHR responded to this risk by:
These mitigation strategies have been successful in managing this risk, and as a result, when CIHR revised its Corporate Risk Profile for 2014-15, the nature of this risk had significantly evolved and the risk response strategy was adapted accordingly. For more information on this risk, please see CIHR's 2014-15 Report on Plans and Priorities.Footnote 42 |
||
Actual Expenditures
| 2013-14 Main Estimates |
2013-14 Planned Spending |
2013-14 Total Authorities Available for Use |
2013-14 Actual Spending (authorities used) |
DifferenceFootnote i (actual minus planned) |
|---|---|---|---|---|
| 967,653,157 | 981,350,017 | 1,001,038,961 | 997,971,988 | 16,621,971 |
|
||||
| 2013-14 Planned |
2013-14 Actual |
2013-14 DifferenceFootnote ii (actual minus planned) |
|---|---|---|
| 390 | 399 | 9 |
|
||
Strategic Outcome, Program(s) and Internal Services |
2013-14 Main Estimates |
Planned Spending | 2013-14 Total Authorities Available for Use |
Actual Spending (authorities used) | ||||
|---|---|---|---|---|---|---|---|---|
| 2013-14 | 2014-15 | 2015-16 | 2013-14 | 2012-13 | 2011-12 | |||
| 1.1 Health Knowledge | 472,958,825 | 473,334,726 | 482,042,223 | 491,680,636 | 474,068,424 | 488,585,954 | 482,927,902 | 470,528,113 |
| 1.2 Health Researchers | 173,268,324 | 172,904,183 | 178,093,133 | 172,515,515 | 173,303,484 | 168,791,689 | 173,088,363 | 182,756,604 |
| 1.3 Health Research Commercialization | 43,822,075 | 53,566,075 | 49,205,349 | 50,219,764 | 58,625,183 | 57,415,525 | 52,306,643 | 55,976,670 |
| 1.4 Health and Health Services Advances | 253,336,423 | 253,765,642 | 252,932,171 | 242,068,700 | 268,478,487 | 256,650,865 | 260,893,758 | 268,011,156 |
| Strategic Outcome Sub-Total | 943,385,647 | 953,570,626 | 962,272,876 | 956,484,615 | 974,475,578 | 971,444,033 | 969,216,666 | 977,272,543 |
| Internal Services Subtotal | 24,267,510 | 27,779,391 | 26,397,475 | 26,367,052 | 26,563,383 | 26,527,955 | 27,836,076 | 31,818,850 |
| Total | 967,653,157 | 981,350,017 | 988,670,351 | 982,851,667 | 1,001,038,961 | 997,971,988 | 997,052,742 | 1,009,091,393 |
Spending and FTE Variance Explanations
The variance between CIHR's Total Authorities of $1,001.0M in 2013-14 and Planned Spending of $981.4M results primarily from $15.0M of funding received through Budget 2013 for CIHR's Strategy for Patient-Oriented Research. In addition, CIHR received additional funding through the Supplementary Estimates for the Business-Led Networks of Centres of Excellence program ($2.6M) and for the Centres of Excellence for Commercialization and Research program ($2.3M). Actual expenditures incurred in 2013-14 are consistent with those incurred in the prior fiscal year.
In 2013-14, CIHR's total expenditures (authorities used) were approximately $998.0M, which was $3.1M less than its Total Authorities due to the fact that CIHR received additional funding through the Supplementary Estimates for the Business-Led Networks of Centres of Excellence program ($2.6M) and for the Centres of Excellence for Commercialization and Research program ($2.3M) after competition results were finalized. CIHR has been authorized to carry forward approximately $2.3M of its 2013-14 unspent authorities to the 2014-15 fiscal year.
No material differences were noted between planned FTEs and actual FTEs in 2013-14.
Alignment of Spending with the Whole-of-Government Framework
| Strategic Outcome | Program | Spending Area | Government of Canada Outcome | 2013-14 Actual Spending |
|---|---|---|---|---|
| 1 A world-class health research enterprise that creates, disseminates and applies new knowledge across all areas of health research | 1.1 Health Knowledge | Social Affairs | Healthy Canadians | 488,585,954 |
| 1.2 Health Researchers | Social Affairs | Healthy Canadians | 168,791,689 | |
| 1.3 Health Research Commercialization | Social Affairs | Healthy Canadians | 57,415,525 | |
| 1.4 Health and Health Services Advances | Social Affairs | Healthy Canadians | 256,650,865 |
| Spending Area | Total Planned Spending | Total Actual Spending |
|---|---|---|
| Economic Affairs | ||
| Social Affairs | 953,570,626 | 971,444,033 |
| International Affairs | ||
| Government Affairs |
Departmental Spending Trend
Departmental Spending Trend Graph
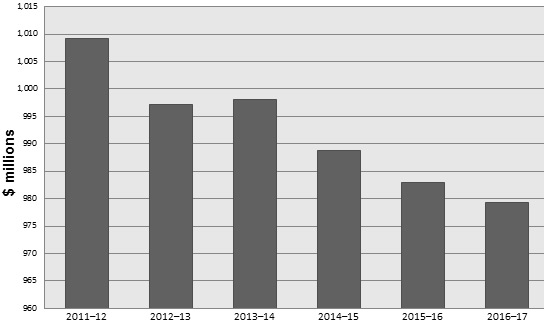
| 2011-12 | 2012-13 | 2013-14 | 2014-15 | 2015-16 | 2016-17 | |
|---|---|---|---|---|---|---|
| Sunset Programs | 0 | 0 | 0 | 0 | 0 | 0 |
| Total Spending | 1,009 | 997 | 998 | 989 | 983 | 979 |
The variance between CIHR's actual total spending of $1,009.1M in 2011-12 and $998.0M in 2013-14 is mainly due to the end of temporary funding received in Budget 2009. Total expenditures are, however, consistent with expenditures incurred in 2012-13, and total planned spending is expected to decrease minimally between 2014-15 ($988.7M) and 2016-17 ($979.3M) as indicated in the graph above. It is important to note, however, that CIHR's share of Tri-Agency programs like the Centres of Excellence for Commercialization and Research (CECR) and the Business-Led Networks of Centres of Excellence (BL-NCE) is not fixed but rather determined by the results of program competitions. Therefore, the expected decrease in planned spending in future years is likely to be offset by additional authorities granted for upcoming CECR and BL-NCE competitions.
Estimates by Vote
For information on the Canadian Institutes of Health Research's organizational Votes and statutory expenditures, consult the Public Accounts of Canada 2014 on the Public Works and Government Services Canada website.Footnote 44
Section II: Analysis of Programs by Strategic Outcome
Strategic Outcome: A world-class health research enterprise that creates, disseminates and applies new knowledge across all areas of health research.
| Health Knowledge | Health Researchers | Health Research Commercialization | Health and Health Services Advances | Internal services |
|---|---|---|---|---|
| 29% | 4% | 1% | 21% | 45% |
CIHR is the Government of Canada's health research funding agency. In 2013-14, CIHR directly supported 13,720 health researchers and trainees in order to improve the Canadian health care system and the health of Canadians. This represents an increase of more than 500 investigators or 4.0% over 2012-13. CIHR supports research through the following programs:
- Health Knowledge
- Health Researchers
- Health Research Commercialization
- Health and Health Services Advances
CIHR uses a peer review process to identify exemplary projects and individuals that merit funding. In 2013-14, approximately 3000 peer reviewers provided their time, without remuneration, and served on over 250 peer review committees to help review over 16,000 applications.
Program 1.1: Health Knowledge
Description
This program aims to support the creation of new knowledge across all areas of health research to improve health and the health system. This is achieved by managing CIHR's open competition and related peer review processes based on internationally accepted standards of scientific excellence.
| 2013-14 Main Estimates |
2013-14 Planned Spending |
2013-14 Total Authorities Available for Use |
2013-14 Actual Spending (authorities used) |
2013-14 Difference (actual minus planned) |
|---|---|---|---|---|
| 472,958,825 | 473,334,726 | 474,068,424 | 488,585,954 | 15,251,228 |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 104 | 114 | 10 |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Health research advances knowledge | Outputs and impacts of CIHR-funded research | Maintain or increase | CIHR met the 2013-14 target by increasing overall expenditures for new and ongoing grants to $473.1M, and by funding 798 new grants compared to $468.7M and 804 new grants in 2012-13. For an example of the impact of this research, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
The variances between planned and actual spending of approximately $15.3M, as well as the FTE variance of 10, are largely due to the high number of quality applications received via the Open Operating Grant ProgramFootnote 45 (OOGP), which resulted in in-year reallocations from other CIHR programs and sub-programs.
In 2013-14, through various competitions and commitments, CIHR provided support for the creation of new knowledge across all areas of health research to improve health and the health system. Through the Open Operating Grant Program, CIHR provided funding to support research in all areas of health and achieved its target of providing approximately 800 new multi-year grants through two open competitions launched in March and September. In 2013-14, CIHR funded 798 new grants for a total committed value of $467.2M over five years. CIHR received and reviewed 4,586 applications through these two grant competitions. Finally, of the grants funded in 2013-14, 208 included a randomized controlled trial component as part of the project.
Through all multi-year investments made in previous competitions, CIHR supported 6,844 researchers by funding 3,661 ongoing grants, which represented $473.1M in expenditures in 2013-14 through the Health Knowledge Program. The annual average grant payment increased from $118,148 in 2012-13 to $129,228 in 2013-14. CIHR has seen a 25% increase in fundable applications since 2010-11. The increase in the average annual grant value has led to a decrease in the total number of applications funded over time.
Researchers supported by CIHR through the Health Knowledge Program made significant contributions to the advancement of knowledge. For example, a new web- and pharmacy-based screening program will identify people with early-stage osteoarthritis and help them get the care they need. Through a partnership between the Arthritis Research Centre of CanadaFootnote 46 (ARC), Arthritis Consumer ExpertsFootnote 47 (ACE) and Shoppers Drug Mart, the program allows people to complete an online screening tool and consult with a pharmacist to detect early signs of osteoarthritis before they experience irreversible damage to their joints. In a pilot project funded by CIHR, patients who tested positive on the screening were then sent to a specialist for a formal medical evaluation. According to Dr. Carlo MarraFootnote 48, professor of pharmacy at UBC and an ARC research scientist, the pharmacists were correct over 93% of the time, showing this is a highly effective tool. As of March 2014, more than 54,000 Canadians had used the online screening tool, demonstrating that the Health Knowledge program is achieving its objectives. It is anticipated that the Reforms of the Open Suite of Programs will have even greater impact once implemented.
Sub-Program 1.1.1: Open Research Grant Program
Description
The Open Research Grant Program provides operating funds to support research proposals in all areas of health research. The Open Operating Grant Program is the largest component of this program. Competitions are typically held each March and September with an open call for research proposals, with no restrictions on areas of research or maximum level of requested funds.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 464,329,864 | 480,420,355 | 16,090,491 |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 102 | 112 | 10 |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Health research is conducted and translated | Number of publications from CIHR-supported research | Maintain or increase | CIHR exceeded the 2013-14 target. The end of grant reports submitted in 2013-14 indicate that CIHR support contributed to the production of 7,037 research publications. This is a large increase over the 2012-13 result of 5,751 and mainly attributable to an improved tracking methodology implemented in 2012. |
| Knowledge Translation activities of funded researchers | Maintain or increase | CIHR met the 2013-14 target. The end of grant reports submitted in 2013-14 showed that 28,192Footnote iii KT activities occurred. This is an increase over the 2012-13 result of 22,069 and mainly attributable to an improved tracking methodology implemented in 2012. | |
|
|||
Performance Analysis and Lessons Learned
For variance explanations, please see Program 1.1 Health Knowledge.
CIHR grants helped to maintain a strong and diverse health research base and continued to demonstrate strong leadership in health research in Canada. CIHR relies on its Research Reporting System (RRS) to demonstrate that health research funded by CIHR is conducted and translated through the number of publications from CIHR-supported research and the knowledge translation activities of its funded researchers.
In 2013-14, the RRS collected information on 1,259 CIHR-funded projects. Approximately 56% were from the Open Research Grant Program. These 707 reports stated that CIHR-supported research resulted in 7,037 journal publications. The data from RRS for the same period also indicated that 28,192 KT activities by researchers occurred, including 15,299 presentations and 4,596 interviews. This substantial increase compared to the previous year can be attributed to the implementation of CIHR's Research Reporting System and is expected to stabilize.
CIHR is committed to ensuring that adequate support is available to applicants, peer reviewers and academic institutions throughout the transition period related to the implementation of its reforms of the Open Suite of Programs and peer review processes. Over the last three years, CIHR has been engaging with the research community on the design and implementation of the open and peer review reforms. In 2013-14, CIHR participated in over 60 meetings with researchers, administrators and partners. These discussions have helped CIHR refine the design and inform the transition to the new programs. For each pilot, training was provided to applicants, reviewers and academic institutions.
CIHR also conducted pilots related to the implementation of its reforms of the Open Suite of ProgramsFootnote 49 and peer review processes, which will allow CIHR to adjust and refine processes and systems in order to best support applicants and reviewers. In 2013-14, pilots were implemented and new knowledge was gained on the process, the complexity of peer review and its information technology enhancements, and on the interdependencies between the various reforms projects. The lessons learned have been used to gain efficiencies in the process and better estimate the timelines for the implementation of the reforms.
Through the Operating Research Grant Program, health research is funded, conducted and translated in all areas of health research.
Sub-Program 1.1.2: Randomized Controlled Trials Program
Description
The Randomized Controlled Trials program supports experiments to evaluate the efficiency and effectiveness of interventions in health or health services by randomly assigning individuals to receive or not receive one or more interventions that are being compared. The results are analyzed by comparing outcomes in the different groups.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 9,004,862 | 8,165,599 | (839,263) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 2 | 2 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| High-quality evidence on the efficacy and effectiveness of interventions in health and health services | Evidence produced by funded trials | All completed trials have demonstrated whether or not interventions under study are effective or not | Not available. As of June 2009, CIHR's Randomized Controlled Trials Program was integrated into the Open Research Grant Program. CIHR funded 208 new and ongoing grants related to Randomized Controlled Trials (RCT) in 2013-14 and received 32 RRS reports on RCT grants. For an example of the evidence resulting from funding trials, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
CIHR's Randomized Controlled Trials Program was integrated into the Open Research Grant Program in 2009. The variance between planned and actual spending is attributable to the fluctuation in the number of quality applications submitted related to randomized control trials from year to year. As a result of this fluctuation, the actual spending was slightly lower for randomized control trials; however the remaining funds were spent on quality applications in the Open Research Grant Program. During 2013-14, CIHR continued to work towards the goals established in the Clinical Trials Summit Action PlanFootnote 50 released in 2012. This action plan outlines a number of recommendations for improving Canada's clinical trial capacity.Footnote 51 Throughout 2013-14, Canada's Research Based Pharmaceutical Companies (Rx&D), HealthCareCAN and CIHR worked to establish the Canadian Clinical Trials Coordinating Centre (CCTCC) to help implement the action plan recommendations. The CCTCC was officially launched in April 2014.
Over the past year, CIHR consulted with the External Advisory Committee on Streamlining Health Research Ethics for advice on developing a standardized application and consent form for clinical trials, and to review existing models of ethics accreditation and harmonization in use across the country.
A good example of funded research trials is that done by Dr. Ilias CagiannosFootnote 52, an uro-oncologist from the Ottawa Hospital who received one year of funding to investigate his hypothesis that cooling the kidney area during the surgical removal of kidney tumors can improve the recovery of kidney function and reduce complications. This project was the first prospective trial to evaluate the clinical impact of renal hypothermia during partial nephrectomy with a potential important impact on surgical technique and patient outcome. These funds were used to further develop the proposal for a subsequent clinical trial.
Program 1.2: Health Researchers
Description
This program aims to build health research capacity to improve health and the health system by supporting the training and careers of excellent health researchers through a competitive peer review process based on internationally accepted standards of scientific excellence.
| 2013-14 Main Estimates |
2013-14 Planned Spending |
2013-14 Total Authorities Available for Use |
2013-14 Actual Spending (authorities used) |
2013-14 Difference (actual minus planned) |
|---|---|---|---|---|
| 173,268,324 | 172,904,183 | 173,303,484 | 168,791,689 | (4,112,494) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 14 | 14 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| A strong and talented health research community with the capacity to undertake health research | Number of graduate trainees in Canada compared to international levels | Maintain or increase international ranking | CIHR met the 2013-14 targets by contributing to the maintenance of the number of PhD graduates in Canada per 100,000 aged 25-29, moving from 226 graduates in 2010 to 234 in 2011.Footnote iv Furthermore, the latest set of complete data for higher education expenditure on research and development (HERD) showed that Canada now leads G-7Footnote 53 countries in postsecondary research investment.Footnote v |
| Number and fields of investigators and trainees funded | Maintain number and diversity (by theme and institute domain) of trainees funded | CIHR did not meet the target: it funded 2,409 investigators and trainees, both new and ongoing, in all areas of research and in all themes in 2013-14Footnote vi, compared to 2,538 in 2012-13. This is mainly related to the winding down of temporary funding received as part of Canada's Economic Action Plan, to fund an additional 200 Canada Graduate Scholarship doctoral scholarships in the 2011 competition. | |
|
|||
Performance Analysis and Lessons Learned
The variance between planned and actual spending of approximately $4.1M is primarily due to an in-year reallocation of forecasted unspent funds to Program 1.1, and the $4.1 M was spent on additional high-quality research proposals through OOGP.
In order to ensure a strong and talented health research community, CIHR tracks the number of graduate trainees in Canada compared to international levels, as well as the number and fields of investigators and trainees funded. In 2013-14, CIHR continued to support diversity by funding excellence in all areas of its mandate, including biomedical; clinical; health systems and services; and social, cultural, environmental and population health research as well as institute-specific research areas.
In 2013-14, CIHR provided funding for 696 new awards (training and salary combined). These awards represent a commitment of $145.1M over the next eight years, which resulted in $31.1M in expenditures in 2013-14.The number of health researchers and trainees supported through the Health Researchers Program by CIHR decreased from 2,538 in 2012-13 to 2,409 in 2013-14.
Through the Health Researchers program, CIHR provides financial support to develop future health researchers at both the investigator and trainee levels in all health-related fields in Canada, enabling them to gain research experience, develop a career in health research and build Canada's health research capacity.
Sub-Program 1.2.1: Salary Support Programs
Description
The Salary Support Programs provide salary support to help new health researchers develop their careers and devote more time to initiating and conducting health research.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 110,918,538 | 110,121,549 | (796,989) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 9 | 9 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Excellent health researchers available to undertake health research | Types and number of health researchers funded | Maintain or increase | CIHR almost met the 2013-14 target by supporting 120 new health researchers through the following salary awards:
|
| Number of fundable applicants who are funded | Maintain or increase | CIHR almost met the 2013-14 target by funding 915 health researchers, a slight decrease compared to the 921 health researchers supported in 2012-13. |
Performance Analysis and Lessons Learned
The Salary Support Programs at CIHR are designed to help new researchers develop their careers and devote more time to research. In 2013-14, CIHR continued to build research capacity to improve health and the health system by investing $110.0M in salary awards to 915 health researchers. A total of 120 new awards were funded, representing a commitment of $93.0M over eight years and $11.2M in expenditures for 2013-14.
Through the Salary Support Programs, CIHR builds research capacity, enhances and consolidates health research by attracting top researchers from Canada and abroad, and retains some of the world's most accomplished and promising researchers in Canada.
Sub-Sub-Program 1.2.1.1: Open Salary Support Programs
Description
The Open Salary Support Programs provide salary support to help new health researchers develop their careers and devote more time to initiating and conducting health research in any area related to health.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 13,910,138 | 13,463,218 | (446,920) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 4 | 4 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Medical/Health, University and Research Institution staff have dedicated time for health research | Amount of work time dedicated to health research | Maintain or increase | Complete data is not available for this measure. In order to demonstrate that promising researchers have dedicated time for health research, CIHR replaced the performance indicators with the number of new investigators supported through the New Investigator Salary Award program, which is designed to enable researchers to reserve time for research and provide outstanding new investigators with the opportunity to develop and demonstrate their independence in initiating and conducting health research. CIHR mostly met this target by maintaining the number of investigators from 235 in 2012-13 to 237 in 2013-14. |
Performance Analysis and Lessons Learned
In 2013-14, CIHR provided salary support to 39 early career researchers, which is equal to 2012-13, thus enabling them to develop and demonstrate their independence in initiating and conducting health research. These new awards represent a commitment of $11.5M over seven years and $1.5M in expenditures for 2013-14.
Sub-Sub-Program 1.2.1.2: Canada Research Chairs Program
Description
The Canada Research Chairs Program aims to attract and retain some of the world's most accomplished and promising researchers. CIHR provides two levels of salary awards (Tier 1 for senior researchers and Tier 2 for junior researchers) that are allocated to universities and research institutions; subsequently researchers are recruited into the "Chair" positions.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 88,608,400 | 88,408,332 | (200,068) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 2 | 2 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| World-class research capacity is enhanced in Canadian universities, research institutes and hospitals through the attraction and retention of excellent researchers | Percentage of Chairs awarded to foreign researchers and expatriates | Greater than or equal to 30% | In 2013-14, CIHR did not meet the target, given that only 12% of active CIHR chairholders were recruited from outside Canada. |
| Enhancement of university role as centres of research excellence - dollar value growth in research centres within 5 years of Chair award | Greater than or equal to 40% | Data for this indicator is not available, given that there is no way to monitor this information. However, in 2012-13, 93% of institutions did indicate that the Canada Research Chairs program is important or very important in their ability to support existing research teams/ research clusters/ research centres. | |
| Percentage of reclaimed Chairs | Less than 5% | In 2013-14, this indicator no longer applies given that the CRC program no longer reclaims vacant chairs. |
Performance Analysis and Lessons Learned
The Canada Research Chairs (CRC) Program is designed to attract and retain some of the world's most accomplished and promising researchers and provides two levels of awards: Tier 1 Chairs are reserved for established researchers, and Tier 2 Chairs support emerging researchers. In 2013-14, CIHR funded 80 new awards for an in-year investment of $8.5M and a total commitment of $71.0M over eight years. This includes 35 new Tier 1 Chairs (for $4.9M) and 45 new Tier 2 Chairs (for $3.6M). The number of new chairs is subject to variability over time; however, there is a negligible variance in total expenditures this fiscal year, with a 1.1% difference between 2012-13 and 2013-14.
Sub-Sub-Program 1.2.1.3: Canada Excellence Research Chairs Program
Description
The Canada Excellence Research Chairs Program targets world-class researchers in the four priority areas of the Government's Science & Technology strategy, including health. These prestigious awards will further encourage the principle of world-class excellence at Canada's post-secondary institutions, and focus resources on strategic priorities that are deemed to be of economic importance to Canada.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 8,400,000 | 8,249,999 | (150,001) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 3 | 3 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| The attraction of the world's most accomplished researchers to help Canada build a critical mass of expertise in areas that align with Science & Technology priority areas | Percentage success in attracting world-class researchers to Canada | Increasing trend | In 2013-14, CIHR met this target as 100% of the CERC were awarded to world-class researchers from outside Canada. |
| Percentage increase in total funding in priority area of CERC at host university | 15% over 5 years | Data for this indicator is not available given there is no way to monitor this information. However, in 2012-13, 83% of institutions reported significant growth over the term of the award in areas of strategic importance to Canada due to CERC. |
Performance Analysis and Lessons Learned
Through the Canada Excellence Research Chairs Tri-Agency Program, CIHR contributed to building and maintaining world-class research by supporting Canadian universities in their efforts to enhance Canada's growing reputation as a global leader in research and innovation. In 2013-14, CIHR awarded $8.2M to six research chairs.
Sub-Program 1.2.2: Training Support Programs
Descriptions
The Training Support Programs provide support and special recognition to PhD, post-PhD or post-health professional degree students who are training in health research areas in Canada or abroad.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 61,985,645 | 58,670,140 | (3,315,505) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 5 | 5 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Highly trained individuals available for health research | Numbers of trainees completing degrees or fellowship programs | Maintain or increase | Complete data is not available for this measure. CIHR is working to implement a training survey similar to the Research Reporting System for grants which would track this information in 2014-15. |
Performance Analysis and Lessons Learned
CIHR contributed to building world-class research capacity by developing, attracting and retaining the best students, both nationally and internationally. In 2013-14, CIHR invested $58.4M through the Training Support Programs in 1,416 trainees in master's, doctoral, postdoctoral or post-health professional degrees in Canada and abroad. A total of 576 new awards were provided, representing a commitment of $52.1M over six years and expenditures of $19.8M in 2013-14. The number of trainees varies from year to year; however, expenditures remained stable given there were $20.3M in expenditures in 2012-13.
CIHR funds many trainees indirectly through other grant programs, in addition to funding trainees through direct stipends. This year, by analyzing administrative data, CIHR was able to determine that some 25% of grant budgets from the Open Operating Grant Program are used to support approximately 960 new graduate students (master's and doctoral) and 440 new postdoctoral fellows every year. This clearly demonstrates that CIHR is developing the capacity of health research in Canada and ensuring a future generation of high-quality health researchers.
Sub-Sub-Program 1.2.2.1: Open Training Support
Description
The Open Training Support programs provide support and special recognition to PhD, post-PhD or post-health professional degree students who are training in any area of health research in Canada or abroad.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 28,556,077 | 25,988,582 | (2,567,495) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 2 | 2 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Highly trained individuals available for health research | Numbers of trainees completing degrees or fellowship programs | Maintain or increase | Complete data is not available for this measure. In order to demonstrate that promising trained individuals are available for health research, CIHR replaced this performance indicator with the number of trainees supported through the CIHR fellowships program, which is designed to support highly skilled, qualified and promising researchers engaged in health research. CIHR met its target by increasing the number of trainees from 455 in 2012-13 to 489 in 2013-14. |
Performance Analysis and Lessons Learned
The variance between planned and actual spending of approximately $2.6M is primarily due to an in-year reallocation of forecasted unspent funds to Program 1.1, and that $2.6M was spent on additional high-quality research proposals through OOGP. The quality of applications fluctuates from year-to-year and as a result, funds are reallocated to fund additional high-quality research applications.
CIHR's Open Training Support sub-sub-program is designed to support highly skilled, qualified and promising researchers and ensure they investigate in health research. In 2013-14, CIHR supported 455 trainees through the CIHR fellowships program. CIHR funded 23 new Banting Postdoctoral Fellowships (including five awarded to foreign citizens), which is equal to the number funded in 2012-13. In total, CIHR supported 49 postdoctoral fellows, 10 of whom were foreign citizens.
Sub-Sub-Program 1.2.2.2: Sir Frederick Banting and Dr. Charles Best Canada Graduate Scholarships Program
Description
The Sir Frederick Banting and Dr. Charles Best Canada Graduate Scholarships Program provides financial support to develop future health researchers at both the masters and doctoral levels in all health-related fields in Canada by supporting them as they obtain their graduate degrees.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 25,079,568 | 24,435,725 | (643,843) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 2 | 2 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Increased capacity to meet demand for highly qualified personnel in the academic, public and private sectors | Percentage of recipients completing degrees | Maintain or increase | In 2013-14, CIHR met this target as 100% of the recipients were completing degrees. |
| Average completion rates vs. general population | Greater than or equal to general population | Complete data is not available for these measures. CIHR is working to implement a training survey similar to the Research Reporting System for grants which would track this information in 2014-15. | |
| Average time to degree completion among recipients vs. general population | Less than or equal to general population |
Performance Analysis and Lessons Learned
CIHR invested $8.5M through the Canada Graduate Scholarships (CGS) in 2013-14 to support the learning and development of its health research trainees by awarding 166 new doctoral awards and 129 new master's awards. This represents a decrease of eight doctoral awards and 51 master's awards since 2012-13 and is mainly attributable to the winding down of temporary funding received as part of Canada's Economic Action Plan to fund an additional 200 CGS doctoral scholarships in the 2011 competition and an additional 400 CGS master's scholarships in the 2010 competition.
Sub-Sub-Program 1.2.2.3: Georges Philias Vanier Canada Graduate Scholarships Program
Description
The Georges Philias Vanier Canada Graduate Scholarships Program will enable Canada to build world-class research capacity by attracting the best doctoral students, both nationally and internationally.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 8,350,000 | 8,245,833 | (104,167) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 1 | 1 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Contribute to making Canada a more attractive destination for international students and making Canada's scholarship initiatives more internationally competitive | Percentage of foreign Vanier candidates put forward by universities | Increasing trend | In 2013-14, CIHR met its target by increasing the percentage of foreign candidates. The percentage of foreign Vanier candidates put forward has been slightly increasing over the years:
|
| Recipients' perception of Canada as an attractive destination for research | 90% of recipients identify Canada as an attractive destination | Complete data is not available for this measure. The Vanier Banting Secretariat is working to implement an end of award report similar to the Research Reporting System for grants, which would track this information in 2014-15. |
Performance Analysis and Lessons Learned
CIHR funded 56 new Vanier Canada Graduate Scholarship awards in 2013-14, an increase from 53 new awards in 2012-13. Thirteen of these awards were given to foreign citizens, which is similar to the number in 2012-13. CIHR supported 166 trainees through this program, 30 of whom are foreign citizens. These recipients are world-class doctoral students who demonstrate a high standard of scholarly achievement in graduate studies, as well as leadership skills.
Program 1.3: Health Research Commercialization
Description
This program aims to support and facilitate the commercialization of health research to improve health and the health system. This is achieved by managing funding competitions to provide grants, in partnership with the private sector where relevant, using peer review processes based on internationally accepted standards of scientific excellence, and by building and strengthening the capacity of Canadian health researchers to engage in the commercialization process.
| 2013-14 Main Estimates |
2013-14 Planned Spending |
2013-14 Total Authorities Available for Use |
2013-14 Actual Spending (authorities used) |
2013-14 Difference (actual minus planned) |
|---|---|---|---|---|
| 43,822,075 | 53,566,075 | 58,625,183 | 57,415,525 | 3,849,450 |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 6 | 6 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Commercial activity - products (patents and intellectual property), companies and employment generated | Outputs and impacts of funded research | Evidence that some work of CIHR-funded researchers resulted in some of these long-term impacts | CIHR met the 2013-14 target with expenditures to support commercialization totalling $57.4M, an increase from $52.3M in 2012-13. A total of 228 grants were funded. For an example of the impact of this research, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
The variance between planned and actual spending of $3.8M is primarily due to additional funding received through Supplementary Estimates for the Business-Led Networks Centres of Excellence program ($2.6M) and for the Centres of Excellence for Commercialization and Research program ($2.3M) after competition results were finalized.
CIHR demonstrated support for and facilitation of the commercialization of health research. In 2013-14, CIHR spent $57.4M on 228 grants in the Health Research Commercialization program, including 93 new grants representing a commitment of $39.7M and expenditures of $15.1M in 2013-14. These figures represent a 9.8% increase over 2012-13 and a 3.7% increase over 2011-12.
CIHR-funded research has had a positive impact on the commercialization of new health products. For example, as a result of commercialization funding from CIHR, Dr. Igor JurisicaFootnote 54 at Princess Margaret Cancer Centre in Toronto developed different tools for the efficient and effective network-based prediction of diagnostic and prognostic cancer signatures. The focus of these tools is to enable network analysis and modelling which can be integrated with cancer profiles. These tools enable the identification of diagnostic and prognostic biomarkers, as well as an understanding of disease initiation and progression, leading to improved cancer treatment. As a result, tools and resourcesFootnote 55 have been commercialized such as NAViGaTOR, I2D, mirDIP, CDIP, and BTSVQ, all of which enable users to interpret integrated cancer profiles and dynamically create relevant models.
Through the Health Research Commercialization program, CIHR supports applied research and collaborations that facilitate commercialization and technology transfer of new discoveries in areas of strategic importance to Canada.
Sub-Program 1.3.1: Research Commercialization Program
Description
The Research Commercialization Program is comprised of a suite of funding initiatives that aim to support the creation of new knowledge, practices, products and services and to facilitate the commercialization of this knowledge.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 19,495,675 | 18,432,233 | (1,063,442) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 2 | 2 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Health research is commercialized more effectively | Outputs and impacts of funded research | Maintain or increase | CIHR met the 2013-14 target with expenditures totalling $18.4M. This is an increase from $13.9M in 2012-13 and $16.8M in 2011-12. A total of 206 grants were funded. Please see the Performance and Analysis and Lessons Learned section below for an example of the impact of this funding. |
Performance Analysis and Lessons Learned
CIHR's Research Commercialization Program provides seed funding for pre-competitive research and development projects; collaborative natural science or engineering and health research projects; support for technology transfer; academic/industry research collaborations; and opportunities for management and entrepreneurial training. Through this program, CIHR encouraged and funded innovative research that will result in solutions to health problems and contribute to economic growth.
To support the creation of new knowledge, practices, products and services and facilitate the commercialization of this knowledge, CIHR spent $18.4M in 2013-14 on 206 grants in the Research Commercialization Program, which is an increase over 2012-13 (164 grants and $13.9M spent). Of these, 87 were new grants ($9.2M) and accounted for half the expenditures in 2013-14.
CIHR-funded research resulted in the commercialization of new health products. An example of the impact of CIHR-funded research is Dr. Ruth MartinFootnote 56 at Western University, who has developed a treatment for swallowing disorders using air pulse therapy, known as Swallowing Air-Pulse Therapy (SWAPT). The treatment helps prompt swallowing in patients by delivering a series of air pulses to the back of the throat. The SWAPT device, which consists of a small control unit with a disposable mouthpiece, has already shown promising results in a clinical trial. Difficulty swallowing (a disorder known as dysphagia) can lead to other serious health issues, including malnutrition and pneumonia. Unfortunately, there are few treatment options for people with dysphagia. Dr. Martin is now working with a commercial partner, Trudell Medical International, to conduct further randomized controlled trials of SWAPT.
In 2013-14, CIHR evaluated the Commercialization programs, and the final report will be published in 2014-15. The results of the evaluation will inform future programming design decisions. CIHR is also consulting with external stakeholders to inform the development of a new commercialization strategy. Findings from the evaluation of the Collaborative Health Research ProjectsFootnote 57, completed in 2013-14, will be used to support the design and delivery of future collaborative interdisciplinary research programs with a commercialization focus.
Through the Research Commercialization Program, CIHR supports the creation of new knowledge, practices, products and services and facilitates the commercialization of knowledge.
Sub-Program 1.3.2: Networks of Centres of Excellence Programs
Description
The Networks of Centres of Excellence grants programs support the networking of centres of research excellence with industrial know-how and strategic investment to turn Canadian research and entrepreneurial talent into economic and social benefits for Canada. The Networks of Centres of Excellence programs are national in scope, multidisciplinary and involve multisector partnerships.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 34,070,400 | 38,983,292 | 4,912,892 |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 4 | 4 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| World-class networks and centres connect leading-edge research with industrial know-how and strategic investment | Significant demonstration of impacts | 100% of networks demonstrate impacts by year 4 | CIHR met this target, and 100% of the networks demonstrated impact. For details concerning the impacts of this sub-program, please see the Performance and Analysis and Lessons Learned section for sub-sub programs 1.3.2.1, 1.3.2.2 and 1.3.2.3 |
| Ratio of partner contributions to NCE funding by networks at year 4 | Greater than or equal to 1 | CIHR exceeded its target by leveraging $1.63 for every $1 invested. |
Performance Analysis and Lessons Learned
For an explanation on the variance between planned and actual spending, please see Program 1.3 Health Research Commercialization.
CIHR funds world-class networks and centres connecting leading-edge research with industrial know-how and strategic investment which is demonstrated through the significant impact and ratio of partner contributions to NCE funding.
The NCE reportFootnote 58 also outlined effectiveness in stimulating partner contributions to innovation research. Partner contributions grew to more than $245M in 2012-13, compared to $150M NCE grant expenditures. The leveraging of partner contributions to NCE funding is $1.63 for every NCE dollar, exceeding the target of greater than or equal to 1.
CIHR's NCE sub-program includes the Networks of Centres of Excellence (NCE); Centres of Excellence for Commercialization and Research (CECR) and the Business-Led Networks of Centres of Excellence (BL-NCE). CIHR administered $39.0M in grants and awards through this sub-program in 2013-14, which is consistent with 2012-13. CIHR funded 22 grants and awards in 2013-14, an increase of 29% from 2012-13.
Sub-Sub-Program 1.3.2.1: Networks of Centres of Excellence Program
Description
The Networks of Centres of Excellence (NCE) grants program provides funds to support the best NCE applications in areas of health research.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 22,589,400 | 22,589,000 | (400) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 2 | 2 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Strong linkages and partnerships created between university, government and industry, and other users (NGOs), resulting in the transfer of knowledge and exploitation of leading-edge research results with economic or societal benefits to Canada | Significant demonstration of impacts by year 4 (i.e. number of patents, licenses, copyrights, number of new products or processes, policies influenced or created, health delivery influenced stemming from network research) | Maintain or increase | CIHR met its target due to the fact that every network demonstrated significant impacts. Overall, Tri-Agency support contributed to 85 patents filed; 10 patents issued; 5 licenses granted; 14 licenses under negotiation; 4 products and innovations; 22 copyrights and 1,598 other knowledge translation and exchange (KTE) activities. |
| Ratio of partner contributions to NCE funding by networks by year 4 | Greater than or equal to 1 | CIHR met its target by leveraging $1 for every $1 invested, which represented $65.6M of contributions from partners versus $66.1M of NCE expenditures.Footnote vii | |
|
|||
Performance Analysis and Lessons Learned
Partnerships fostered by the Tri-Agency Networks of Centres of Excellence (NCE) Program ensure that Canadian research is turned into social and economic benefits for Canadians. The 2012-13 NCE report (which is the most recently published report) outlined that the NCE resulted in 398 new companies and the publication of 2,841 peer-reviewed articles, clearly demonstrating the advancement of knowledge and an impact on the economy.
The NCE report also outlined effectiveness in stimulating partner contributions to innovation research. Partner contributions grew to more than $245M in 2012-13, compared to $150M NCE grant expenditures. The leveraging of partner contributions to NCE funding is $1.63 for every NCE dollar, exceeding the target of greater than or equal to 1.
Overall, Tri-Agency investment in the NCE programs demonstrates its ability to turn Canadian research and entrepreneurial talent into economic and social benefits for Canada.
A review of relevance and effectiveness of the Networks Centres of Excellence Program was completed for this sub-program in 2012-13. The evidence gathered for this review demonstrates that the NCE program's investments in a networking approach to research have enhanced research, development and innovation in areas targeted by the funded networks.
Sub-Sub-Program 1.3.2.2: Business-Led Networks of Centres of Excellence Program
Description
The Business-Led Networks of Centres of Excellence Program aims to mobilize Canada's research talent in the academic, private, public and not-for-profit sectors and apply it to developing the economy and improving the health and quality of life of Canadians. The program funds large-scale collaborative networks to perform research to support private sector innovation in order to deliver economic, health, social and environmental benefits to Canada.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 1,737,000 | 4,340,132 | 2,603,132 |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 1 | 1 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Strengthened public-private sector collaborations, to address significant research challenges meeting business needs and to accelerate the commercialization of leading-edge technologies, goods and services in priority areas where Canada can significantly advance its competitive advantage | Significant demonstration of short-term impacts by year 4 (i.e. business applications/proposals with a clear path to market) | 100% of networks by year 4 | CIHR met its target due to the fact that 100% of the funded networks demonstrated significant short-term impacts. |
| Ratio of partner contributions to business-led funding for networks by year 4 | Greater than or equal to one | CIHR exceeded the target by leveraging $1.52 for every $1 invested, which represented $23.2M of contributions from partners versus the $15.3M of Business-led NCE expenditures.Footnote viii | |
|
|||
Performance Analysis and Lessons Learned
For an explanation on the variance between planned and actual spending, please see Program 1.3 Health Research Commercialization.
In 2013-14, the business-led NCE networks demonstrated significant leveraging of $1.52 for every $1 that CIHR invested. There was a total of $23.2M in contributions from partners, in addition to the $15.3M invested.
In 2013-14, 100% of the funded business-led NCE networks demonstrated significant short-term impacts. For example, the Quebec Consortium for Drug DiscoveryFootnote 59 has demonstrated that over 70% of completed projects have resulted in the execution of a licence by the industry partners.
Sub-Sub-Program 1.3.2.3: Centres of Excellence for Commercialization and Research Program
Description
The Centres of Excellence for Commercialization and Research Program supports, on an open and competitive basis, the operation of research and commercialization centres that bring together people, services and research infrastructure to position Canada at the forefront of breakthrough innovations. The goal of the program is to support internationally recognized centres of commercialization and research expertise in priority areas in order to deliver economic, health, social and environmental benefits to Canada.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 9,744,000 | 12,054,160 | 2,310,160 |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 1 | 1 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Centres with strong commercialization orientation are created that will become self-sufficient | Number of self-sufficient centres | Maintain or increase | In 2013-14, the target was ambitious given that the different centres are at various phases of implementation, and it is too early to have accurate data. However, partial data showed that from 2008 and 2009 competitions, 17 centres were funded; 7 were granted extended funding in subsequent competitions based on a rigorous review of their business plans and an expectation that they will be self-sustaining long after grant. |
| Creation of centres with strong research orientation, yielding significant public benefits within the funding period | Percentage of centres with significant public benefits or research outcomes (for centres with a research focus) | Maintain or increase | CIHR met its target as 100% of the funded centres have significant public benefits or research outcomes. |
Performance Analysis and Lessons Learned
For an explanation on the variance between planned and actual spending, please see Program 1.3 Health Research Commercialization.
A 2012 evaluation of the Centres of Excellence for Commercialization and Research Program recipients funded through the 2008-09 competition highlighted that a longer timeframe is required for research benefits to be realized. As noted above, a new commercialization strategy that will address challenges of commercialization is in the early stages of discussion at CIHR.
An example of the public benefit from the Centres of Excellence for Commercialization and Research Program is the Prostate Centre's Translational Research Initiative for Accelerated Discovery and Development (PC-TRIADDFootnote 60). This initiative is helping OncoGenex (a company spun off from the Vancouver Prostate Centre) to reduce the risks and costs associated with getting new drugs approved by providing critical preclinical development and access to a national clinical trial network.
Program 1.4: Health and Health Services Advances
Description
Through the competitive peer review process based on internationally accepted standards of scientific excellence, this program aims to support the creation of new knowledge in strategic priority areas and its translation into improved health and a strengthened health system.
| 2013-14 Main Estimates |
2013-14 Planned Spending |
2013-14 Total Authorities Available for Use |
2013-14 Actual Spending (authorities used) |
2013-14 Difference (actual minus planned) |
|---|---|---|---|---|
| 253,336,423 | 253,765,642 | 268,478,487 | 256,650,865 | 2,885,223 |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 88 | 85 | (3) |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Translation and use of health research takes place | Impacts of CIHR- funded research | Anecdotal evidence that work of CIHR-funded researchers resulted in some long-term impacts | For details concerning the anecdotal evidence of CIHR-funded researchers of this program, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
The variance between planned and actual spending of approximately $2.9M is due to additional funding allocated to CIHR's Strategy for Patient-Oriented Research in Budget 2013.
CIHR provides funding to researchers for emergent and targeted research that responds to the changing health needs and priorities of Canadians across all aspects of health. In 2013-14, CIHR supported the creation of new knowledge in strategic priority areas through both signature and institute initiatives.
In 2013-14, CIHR continued with the implementation of the Strategy for Patient-Oriented Research (SPOR), which is a signature initiative and a priority for the Government of Canada that received additional funding in the 2014 Economic Action Plan. Two core elements of the strategy, SPOR networks and Support for People and Patient-Oriented Research and Trials (SUPPORT) Units, both made significant progress.
The 2014 Economic Action Plan also renewed investments in health research to tackle the growing onset of dementia and related illnesses. This new funding allows multidisciplinary collaborative research teams from across Canada to engage in innovative and impactful research.
The success of the institute and signature initiatives is reliant on partner collaborations. The design of the competition for the Canadian Consortium on Neurodegeneration in AgingFootnote 61 (CCNA) included a partners' forum to facilitate integrated knowledge translation. This provided a platform for researchers, partners and other stakeholders to exchange ideas and contribute to shaping the CCNA's research agenda from the early stages of its inception. This approach will be used by CIHR for other large-scale funding opportunities.
In 2013-14, the Research Reporting System received 500 reports related to strategic grant programs. Data from these reports indicated that the research supported by CIHR grants resulted in the production of 1,782 journal publications. Furthermore, these CIHR-supported research projects produced an additional 10,598 KT activities, including 5,671 presentations and 2,499 interviews.
The Health and Health Services Advances program demonstrates that CIHR is able to create new knowledge which is translated and used to improve the health of Canadians, create more effective health services and products and strengthen the Canadian health care system through targeted investment in priority areas.
Sub-Program 1.4.1: Institute Strategic Initiatives
Description
The Institute Strategic Initiatives program is led by CIHR's 13 Institutes and funds grants and awards to support research and researchers in priority areas to address strategic health opportunities, threats and challenges to Canadians. The Institutes identify these strategic priority research areas in consultation with stakeholders from government, health care, patient and community groups, and industry. Proposals are solicited from researchers by issuing a Request for Applications outlining the specific theme/area where research is needed. Applications are peer reviewed using criteria specific to the funding opportunity.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 233,108,109 | 236,940,907 | 3,832,798 |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 81 | 78 | (3) |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Health research is conducted, translated and used in priority areas | Outputs and impacts of funded research and success against objectives | Maintain or increase | CIHR met the 2013-14 target with expenditures totalling $226.2M in strategic initiatives, which is consistent with the $226.3M in 2012-13. For details on the impact of this research, please see the Performance Analysis and Lessons Learned section. |
Performance Analysis and Lessons Learned
In an effort to achieve CIHR's goal of addressing health and health system research priorities, CIHR created signature and strategic initiatives to attain greater focus and impact from our strategic investments.
In 2013-14, CIHR spent $226.2M on 2,186 grants related to the sub-program Institute Strategic Initiatives. A total of 1,302 new grants representing commitments of $307.4M over seven years and $70.6M in 2013-14 expenditures were approved. This is a slight decrease of 124 grants or 5.4% from 2012-13, when CIHR supported 2,310 grants. The decrease in the number of grants is directly attributable to the increase in the annual average grant payment from $97,982 in 2012-13 to $103,492 in 2013-14.
In 2013-14, CIHR identified the need for a clear and consistent way of developing, reviewing and analyzing new proposals for large-scale signature initiatives which would outline the governance, objectives and performance measures to improve program monitoring and reporting. As a result, CIHR developed and approved a governance strategy for CIHR-funded major initiatives in 2013-14, and as part of the implementation, all major initiatives previously approved are in the process of developing their governance structure and updating their performance measurement strategy accordingly. This will ensure a consistent approach across CIHR that will improve monitoring and reporting and inform decision making.
The Institute Strategic Initiatives sub-program supports research and researchers in priority areas to address strategic health opportunities, threats and challenges to Canadians.
Sub-Sub-Program 1.4.1.1: Strategic Initiatives Program
Description
The Strategic Initiatives Program funds grants and awards to support research and researchers in priority areas to address the health priorities of Canadians. The Institutes and other branches at CIHR, where relevant, identify these strategic research priorities in consultation with stakeholders from government; health care, patient and community groups; and industry.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 156,041,482 | 157,714,939 | 1,673,457 |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 54 | 50 | (4) |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Effective domestic and international health research agendas as well as alliances and partnerships in areas related to the mandate of each Institute | Evidence of Institute's emerging leadership within the Canadian research and research user community, including examples of institute innovation in identifying and responding to national and international health threats and opportunities. | Evidence that CIHR Institutes emerged as leaders within the Canadian research community | In 2013-14, CIHR demonstrated leadership on many occasions. For example, in partnership with the Indian Council of Medical Research, CIHR is funding research to create knowledge and build capacity in understanding the causes and prevention of childhood obesity and thereby reducing the burden of obesity and its chronic disease effects in both India and Canada. Co-led by Canadian and Indian researchers, the program will foster a long-term institutional partnership involving basic and population health researchers from both countries, as well as training opportunities for PhD students and postdoctoral fellows. |
Performance Analysis and Lessons Learned
CIHR and partners invested $33M in 12 Community-Based Primary Health Care (CBPHC) innovation teams to tackle pressing challenges (related to chronic disease prevention and management, and inequities in access to care for vulnerable populations) and develop evidence-informed solutions to improve CBPHC for Canadians.
In order to better engage First Nations, Inuit, Métis and Urban Aboriginals, and as part of the Pathways to Health Equity for Aboriginal Peoples signature initiative, CIHR is providing $3M over five years to three Aboriginal organizationsFootnote 62 : the Assembly of Manitoba Chiefs, the National Association of Friendship Centres and the Native Women's Association of Canada. This will enable them to become partners for engagement and knowledge exchange in order to stimulate research and enhance understanding of how to create better preventive health services, healthier communities, and health equity for Aboriginal peoples.
Two evaluations were completed for this sub-sub-program: the evaluation of the Regenerative Medicine and Nanomedicine InitiativeFootnote 63 (RMNI); and the evaluation of the Development Innovation Fund: Health. The findings and conclusions from the evaluation of RMNI will be used to inform the design of future funding initiatives and mechanisms, particularly in regard to promoting the integration of CIHR institute and strategic initiatives within the operational framework being developed through the reforms of the Open Suite of Programs.
Sub-Sub-Program 1.4.1.2: Institute Support Grants Program
Description
The Institute Support Grants Program provides funding support to enable institute activities such as the development of strategic health research priorities and research partnerships with government; health care, patient and community groups; and other stakeholders. These priorities and partnerships, along with Institute-organized conferences, seminars and workshops, enable the Institutes to support the growth of the research community, as well as plan and launch their various strategic priority research grants and awards programs.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 13,000,000 | 13,000,000 | - |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 1 | 1 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Effective domestic and international health research agendas as well as alliances and partnerships in areas related to the mandate of each Institute | Evidence of Institute's emerging leadership within the Canadian research and research user community, including examples of institute innovation in identifying and responding to national and international health threats and opportunities. | Evidence that CIHR Institutes emerged as leaders within the Canadian research community | In 2013-14, CIHR provided each of its 13 Institutes with an institute support grant of $1.0M, primarily to support activities that facilitate and develop national and international health research networks linking the Institutes to their respective research communities. For evidence that CIHR emerged as leaders within the Canadian research community, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
CIHR provided each Institute with $1M to support activities that facilitate and develop national and internal health research networks linking the Institutes to their respective research communities. The evidence of the Institutes' leadership is reported on their respective websites.Footnote 64 Furthermore, in 2011 during the last international reviewFootnote 65, the international review panel assessed the mandate-specific reviews of each Institute. The panel noted that the Institutes were fulfilling their mandates and contributing to the improvement of the health of Canadians by catalyzing research and creating productive research partnerships with both investigators and other Institutes.
Sub-Sub-Program 1.4.1.3: HIV/AIDS Research Initiative
Description
The HIV/AIDS Research Initiative is a targeted investment that supports four research streams - biomedical and clinical research, health services and population health research, community-based research, and the Canadian HIV Trials Network.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 25,025,244 | 25,064,250 | 39,006 |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 9 | 9 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Health research advances knowledge and responds to objectives of the HIV/AIDS Research Initiative | Outputs and impacts of funded research and success against objectives | Maintain or increase | CIHR grant and award expenditures totaled $23.9M, which is consistent with the $23.5M in 2012-13. For details concerning impacts of funded research and success against objectives of this program, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
The CIHR HIV/AIDS Research Initiative delivers the research components of two horizontal Government of Canada initiatives on HIV/AIDS: the Federal Initiative to Address HIV/AIDS in Canada and the Canadian HIV Vaccine Initiative.
The evaluation of the CIHR Clinical Trials Network in HIV/AIDS programFootnote 66 concluded that it has made significant contributions to supporting research infrastructure within Canada, building research capacity and creating knowledge in the field of HIV/AIDS clinical trials. The findings and recommendations from the evaluation outlined a number of refinements to both the relevance and design of the CTN program. The evaluation was used to enhance the 2013-14 re-launch of the CIHR Clinical Trials Network in HIV/AIDS program. Following international peer review, the long-standing CIHR Canadian HIV Trials NetworkFootnote 67 was approved for an additional five years, an investment worth nearly $23M.
The HIV/AIDS Research InitiativeFootnote 68 re-launched another infrastructure program in 2013-14, the Centres for Population Health and Health Services Research, based on a 2012-13 mid-term program evaluation. This program is designed to support centres that will help solve current and emerging challenges with the ongoing transmission of HIV, as well as for people living with HIV in Canada.
CIHR developed a new partnership with the Bill & Melinda Gates Foundation to further HIV vaccine research. In addition, two out of four applications received funding as Canada's contribution to international efforts to find a cure for HIV.Footnote 69
The horizontal evaluation of the Federal Initiative to Address HIV/AIDSFootnote 70 in Canada, led by the Public Health Agency of Canada, found that the initiative is making solid progress to enhance individual and organizational capacity to address HIV/AIDS. Findings from the CIHR CTN evaluation were used as evidence that the initiative has led to the creation of new and valuable knowledge to better understand and combat HIV/AIDS in Canada and abroad.
Sub-Sub-Program 1.4.1.4: Pandemic Preparedness Strategic Research Initiative
Description
The Pandemic Preparedness Strategic Research Initiative identifies strategic research priorities and supports pandemic preparedness research. This targeted investment aims to identify strategies to prevent or mitigate a pandemic outbreak as well as methods and procedures to control disease spread (both human to human and from animals to humans) and to treat affected individuals.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 1,509,000 | 1,509,000 | - |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 1 | 1 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Health research advances knowledge and responds to objectives of the Pandemic Preparedness Strategic Research Initiative | Outputs and impacts of funded research and success against objectives | Maintain or increase | CIHR met the 2013-14 target with expenditures for the Pandemic Preparedness Strategic Research Initiative totalling $1.5M, which is consistent with the $1.4M in 2012-13. The Pandemic Preparedness Strategic Research Initiative was sunset in 2012-13, which explains the decrease in expenditures from previous years. For details concerning impacts of funded research and success against objectives of this program, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
The evaluation of the Pandemic Preparedness Strategic Research InitiativeFootnote 71, completed 2013-14, found that the initiative rapidly responded to the need to improve pandemic preparedness and response in Canada. Findings from the evaluation indicate that the initiative was effective in developing collaborations and partnerships between researchers, health service providers and policy makers, and in building capacity in Canada in the area of pandemic research. The evaluation also identified a number of lessons learned that have the potential to inform future CIHR funding initiatives, particularly with regard to targeted funding mechanism designed to create knowledge and increase research capacity in a specific area.
Sub-Sub-Program 1.4.1.5: National Anti-Drug Strategy Treatment Research Initiative
Description
The National Anti-Drug Strategy Treatment Research Initiative is a targeted investment to support grants for strategic research that will help develop and evaluate drug treatment models and approaches, as part of the strategy's treatment action plan.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 935,000 | 869,578 | (65,422) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 1 | 1 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| World-class research related to improving the effectiveness and informing policy and program development of and for addiction treatments (for youth, First Nations and Inuit peoples) | Outputs and impacts of funded research and success against objectives | Evidence of impacts of NADS funding | CIHR met its target by maintaining the expenditures of $0.87M in 2013-14, which is consistent with the $0.94M in 2012-13. In 2013-14, CIHR funded 3 ongoing team grants and 10 new grants as part of the Canadian Research Initiative in Substance Misuse. In 2012-13, CIHR supported the 3 ongoing team grants and 1 one-year grant. For details concerning impacts of funded research and success against objectives of this program, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
In 2013-14, the success and the impact of the investments in the National Anti-Drug Strategy Treatment Research Initiative are reflected in the work accomplished through the three team grants. Dr. Benedikt FischerFootnote 72 and his team have published a series of papers characterizing the issue of prescription opioid abuse in Canada, including a studyFootnote 73 of changes in prescription opioid dispensing trends following recent policy changes and interventions. In the study, Dr. Fischer shows that in select provinces where prescription opioid abuse-related interventions were applied, previous increasing trends in dispensing were reversed. This change could inform the use of similar interventions in other provinces.
The group of Dr. Thomas BrownFootnote 74 has done research to identify individuals at risk for recidivism of driving while impaired. Using neurocognitive criteria, they have been able to subtype offenders and test specific interventions to reduce recidivism. Dr. Brown and his team are working with the Société de l'assurance automobile du Québec to apply the screenings and interventions they have developed.
The team led by Dr. Kathryn GillFootnote 75 is studying the process of implementing a program of addiction and mental health intervention into primary health care settings. Primary health workers feel they do not have the right tools to screen and provide interventions and treatment to clients with addiction and co-morbid conditions. Working with the service providers, Dr. Gill's team has developed specific modules for interventions in primary health care settings and is studying the knowledge translation process for successful replication in other settings.
Sub-Sub-Program 1.4.1.6: Strategy for Patient-Oriented Research Initiative
Description
The Strategy for Patient-Oriented Research Initiative will effectively bridge the gap between basic health research discoveries and their application to the understanding, treatment and prevention of human disease. It will foster a culture of inquiry at all levels of the health system by supporting people, infrastructure and programs devoted to leading-edge patient-oriented research, with the aim of ultimately improving health outcomes.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 27,345,461 | 30,633,563 | 3,288,102 |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 9 | 10 | 1 |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Health research advances knowledge and responds to objectives of the Strategy for Patient-Oriented Research Initiative | Outputs and impacts of funded research and success against objectives | Evidence of impacts of SPOR funding | In 2013-14, the implementation of the Strategy for Patient-Oriented Research (SPOR) moved forward within all of the critical components of the initiative: SUPPORT units, SPOR networks, capacity building, improving the environment for clinical research, and patient engagement. |
Performance Analysis and Lessons Learned
The variances between planned and actual spending of $3.3M, as well as the variance in FTEs, are due to additional grant funding announced in Budget 2013 (and received via the Supplementary Estimates) to fund CIHR's Strategy for Patient-Oriented Research.
Four SUPPORT units, in Alberta, Manitoba, the Maritimes and Ontario, received funding in 2013-14 and were being implemented. These SUPPORT units have started to establish data linkages, to expand data access, to provide research and methodological advice to support real-world clinical trials, and to provide training to researchers in knowledge translation and patient engagement.
CIHR, in collaboration with the Graham Boeckh Foundation through a partnership entitled Transformational Research in Adolescent Mental HealthFootnote 76 (TRAM), announced funding for the SPOR Network in Youth and Adolescent Mental Health, the first of a series of SPOR networks. TRAM received 54 applications from groups across Canada, and 19 groups were invited to participate in the network development process and TRAM's Strengthening Workshop. The submission deadline for full applications was March 2014; following peer review, the award to the top-ranked network was announcedFootnote 77 in June.
Another SPOR network, the Network in Primary and Integrated Health Care Innovations, was designed to be a network of networks that builds on provincial and territorial networks and national assets to support evidence-informed transformation and delivery of more cost-effective and integrated health care to improve clinical, population health, health equity, and health system outcomes. A total of $898,220 was awarded to 11 provincial/territorial networks to facilitate the development of the Network in Primary and Integrated Health Care Innovations.
A workshop on SPOR capacity developmentFootnote 78 was held, with participants including patients, National Alliance of Provincial Health Research Organizations (NAPHRO) members, SUPPORT unit representatives, experts in patient-oriented research, as well as CIHR Scientific Directors and senior management. Based on a common understanding of patient-oriented research and its capacity challenge, the participants explored a shared approach to addressing the patient-oriented research capacity challenge and provided knowledge that will inform a common vision for a SPOR capacity-development strategy.
In 2013-14, a patient engagement workshopFootnote 79 was held, convening a cross-representation of Canadian patients and patient engagement experts from relevant partner organizations to share experiences, knowledge and lessons learned that can be applied to SPOR. Participants explored and deliberated on how best to engage/inform/interact with patients along the entire research process. A SPOR patient engagement framework was subsequently developed based on the recommendations from the workshop and further consultations with various stakeholders.
Sub-Sub-Program 1.4.1.7: Drug Safety and Effectiveness Network Initiative
Description
The Drug Safety and Effectiveness Network Initiative will link centres of excellence in post-market pharmaceutical research across Canada to facilitate targeted research to increase knowledge about the real-world safety and effectiveness of drugs.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 9,251,922 | 8,149,577 | (1,102,345) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 6 | 6 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Health research advances knowledge and responds to objectives of the Drug Safety and Effectiveness Network | Outputs and impacts of funded research and success against objectives | Maintain or increase | CIHR met the 2013-14 target by maintaining the expenditures of $7.8M for the Drug Safety and Effectiveness Network (DSEN). For details concerning impacts of funded research and success against objectives of this program, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
The variance between planned and actual spending of $1.1M in 2013-14 is due to the annual unpredictability and variability of research queries brought forward by DSEN stakeholders.
In addition to ongoing funding committed in previous fiscal years, DSEN provided rapid funding for targeted research grants to DSEN research teams in order to respond to specific queries. In 2013-14, DSEN funded six projects through its rapid funding process, an investment of $1.3M over three fiscal years. DSEN also launched a new rapid funding opportunity to fund an additional $0.2M over two fiscal years beginning in May 2014.
To support research capacity development in post-market drug safety and effectiveness, DSEN invested $400,000 over four fiscal years in the career development of two doctoral and two postdoctoral trainees, starting in 2014-15.
The Drug Safety and Effectiveness NetworkFootnote 80 was established to increase evidence on drug safety and effectiveness available to regulators, policy makers, and health care providers and patients, and to increase the capacity within Canada to undertake high-quality post-market research in this area. At year end, 17 DSEN queries were completed (representing a 143% increase in activity from 2012-13). DSEN shared the results with the federal regulator or federal/provincial decision makers; 23 queries are currently under research, and six are in development.
Sub-Program 1.4.2: Knowledge Translation Programs
Description
The Knowledge Translation (KT) Programs consist of a suite of funding opportunities that aim to support the synthesis, dissemination, exchange and ethically sound application of knowledge in any area of health research. These programs support the science of KT, capacity development in KT science, and integrated KT-collaborative research which involves researchers and knowledge users working together to address relevant research questions and to exchange and apply knowledge to help solve health and health system problems.
| 2013-14 Planned Spending |
2013-14 Actual Spending |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 20,657,533 | 19,709,958 | (947,575) |
| 2013-14 Planned |
2013-14 Actual |
2013-14 Difference (actual minus planned) |
|---|---|---|
| 7 | 7 | - |
| Expected Results | Performance Indicators | Targets | Actual results |
|---|---|---|---|
| Health research is translated more effectively | Number of knowledge translation activities supported by CIHR (and its partners, where relevant) or resulting from CIHR activities (e.g. synthesis papers, briefs, participation in policy task forces, etc.) | Maintain or increase | In 2013-14, CIHR-supported research projects produced an average of 14.2 KT activities per grant per year of CIHR support, which is a decrease compared to the average of 16.9 in 2012-13. Although CIHR did not meet the target, the decrease is related to improved data collection, which has increased the number of relevant grants.Footnote ix |
| Number of stakeholders engaged in CIHR KT | Maintain or increase | In 2013-14, 507 of 948 CIHR-supported project end of grant reports indicated that they involved at least one stakeholder group in KT activities. This is the first year data is available since the evaluation. Trend information will be available in future years.Footnote x | |
| Outputs and impacts of funded research and success against objectives | Maintain or increase | CIHR did not meet the 2013-14 target. CIHR funded 532 knowledge translation grants, approximately the same as the 538 grants funded in 2012-13 but a decrease from the 671 funded in 2011-12. For details concerning the impacts of this sub-program, please see the Performance Analysis and Lessons Learned section below. | |
Performance Analysis and Lessons Learned
CIHR plays a key role in ensuring that health research is translated more effectively. CIHR's integrated knowledge translation programs include Partnerships for Health System ImprovementFootnote 81, Knowledge to ActionFootnote 82 and Knowledge synthesis.Footnote 83 Elements of the new Open Suite of Programs have begun to be piloted in 2013-14 to ensure that there are no barriers to applications that use an integrated knowledge translation approach to research in the future.
In 2013-14, CIHR supported 532 knowledge translation grants and spent $18.8M, a decrease from $21.9M in 2012-13. A number of KT programs were sunset or funds were redirected to other strategic initiatives, resulting in an overall reduction in the number of grants (from 671 to 532) between 2011-12 and 2013-14. Two examples that account for the bulk of the difference are:
- The KT supplement program, which was a pilot program that sunset in 2011-12, and accounts for 68 grants; and
- A reduction in the number of planning and dissemination grants offered as new strategic priorities emerged, resulting in a decrease of 65 grants.
In 2013-14, CIHR completed an evaluation of its Knowledge Translation Funding ProgramFootnote 84 and found that the program has performed well against CIHR's measures of success. The evaluation identified positive impacts and provided recommendations on how these types of impacts could be maintained as CIHR reduces the complexity of its funding programs, minimizes applicant and peer reviewer burden, and maintains its commitment to research excellence through the reforms to the Open Suite of Programs. Specifically, the findings and recommendations informed the decision by CIHR to integrate KT funding opportunities into the proposed Foundation and Project schemes within the Open Suite of Programs, into CIHR's strategic initiatives as well as into the details of the open and peer review reforms (e.g. guidelines for assessing KT, baselines for measuring KT and changes to the peer review process).
CIHR has demonstrated its expertise in translating health research effectively. Indeed, the end of grant reports submitted in 2013-14 indicate that 96.9% of CIHR-supported projects have resulted in at least one research outcome among 16 pre-identified by CIHR in the RRS, including direct cost savings, product licenses, patents, new or advanced research methods, and new or advanced theories.
Internal Services
Description
Internal Services are groups of related activities and resources that are administered to support the needs of programs and other corporate obligations of an organization. These groups are: Management and Oversight Services; Communications Services; Legal Services; Human Resources Management Services; Financial Management Services; Information Management Services; Information Technology Services; Real Property Services; Materiel Services; Acquisition Services; and Other Administrative Services. Internal Services include only those activities and resources that apply across an organization and not to those provided specifically to a program.
| 2013-14 Main Estimates |
2013-14 Planned Spending |
2013-14 Total Authorities Available for Use |
2013-14 Actual Spending (authorities used) |
2013-14 Difference (actual minus planned) |
|---|---|---|---|---|
| 24,267,510 | 27,779,391 | 26,563,383 | 26,527,955 | (1,251,436) |
| Planned 2013-14 | Actual 2013-14 | Difference 2013-14 |
|---|---|---|
| 178 | 180 | 2 |
Performance Analysis and Lessons Learned
The Internal Services program ensures smooth and efficient processes and administers the needs of its programs and other corporate obligations.
As part of its mandate, in 2013-14 CIHR began a review of the institute model. Aligned with this recommendation, an internal working group (consisting of the 13 Scientific Directors) and an external working group (consisting of highly respected health experts with knowledge of CIHR and a broad understanding of health research) were created and tasked with developing recommendations regarding the model, role and slate of Institutes for consideration by CIHR's Governing Council in August 2014.
In 2013-14, CIHR also began to develop a new strategic plan, Roadmap II, which will build and expand on achievements reached with the current strategic plan, Health Research Roadmap. CIHR has made significant progress towards its goals, and Roadmap II will represent CIHR's continued vision to capture excellence and achieve better health outcomes for Canadians.
In an effort to improve the reporting and monitoring of performance, CIHR undertook and approved a new Management Resources and Results Structure, which was implemented on April 1, 2014. As part of this process, CIHR updated its strategic outcome, and simplified and improved its Program Alignment Architecture, as well as the relevant Performance Management Framework. Additionally, a new Performance Measurement Regime was developed and approved to enable CIHR to report not only on the outputs of its funding but also on the transformational impact of the research it funds. The new PM Regime will be implemented in 2014-15.
A main source of data for the new Performance Measurement Regime is the Research Reporting System, which underwent a thorough review in the 2013-14 fiscal year. This review has resulted in enhancements which will be rolled out over the next few years in order to improve data collection and reduce the respondent burden.
Finally, as part of the evaluation of the Open Operating SupportFootnote 85 Grant in 2012-13, a recommendation to award larger grants with longer terms for the leading investigators nationally was proposed. As a result, CIHR launched the new Foundation Scheme in 2013-14 as part of reforms of its Open Suite of Programs and peer review processes. This new program will provide long-term support to health research leaders with demonstrated track records of success, and will further allow researchers to spend less time writing grant applications so that more time can be spent conducting research.
The Internal Services program ensures organizational excellence through smooth and efficient processes that monitor performance and inform decision making.
Section III: Supplementary Information
Financial Statements Highlights
| 2013-14 Planned Results |
2013-14 Actual |
2012-13 Actual |
Difference (2013-14 actual minus 2013-14 planned) | Difference (2013-14 actual minus 2012-13 actual) | |
|---|---|---|---|---|---|
| Total expenses | 999,947,000 | 1,008,884,000 | 1,011,924,000 | 8,937,000 | (3,040,000) |
| Total revenues | 15,991,000 | 6,854,000 | 16,009,000 | (9,137,000) | (9,155,000) |
| Net cost of operations before government funding and transfers | 983,956,000 | 1,002,030,000 | 995,915,000 | 18,074,000 | 6,115,000 |
| Departmental net financial position | (1,135,000) | (2,203,000) | 342,000 | (1,068,000) | (2,545,000) |
Total expenses were approximately $8.9M higher than planned, due primarily to additional funding announced in Budget 2013 for CIHR's Strategy for Patient-Oriented Research initiative. Total expenses incurred in 2013-14 were consistent with those incurred in the prior fiscal year. Total revenues fluctuate annually as they depend exclusively on the timing of receiving partner funding and disbursing that partner funding to health researchers.
Higher than planned total expenses of $8.9M in 2013-14 and lower than planned total revenues of $9.1M resulted in net cost of operations before government funding and transfers being approximately $18.1M higher than planned for the fiscal year. However, net cost of operations before government funding and transfers increased by only $6.1M (or 0.6%) over the prior fiscal year, as CIHR's operations are very consistent on a year-over-year basis.
| 2013-14 | 2012-13 | Difference (2013-14 minus 2012-13) |
|
|---|---|---|---|
| Total net liabilities | 14,395,000 | 12,084,000 | 2,311,000 |
| Total net financial assets | 11,196,000 | 9,329,000 | 1,867,000 |
| Departmental net debt | 3,199,000 | 2,755,000 | 444,000 |
| Total non-financial assets | 996,000 | 3,097,000 | (2,101,000) |
| Departmental net financial position | (2,203,000) | 342,000 | (2,545,000) |
Total net liabilities ($14.4M) and total net financial assets ($11.2M) both increased in 2013-14 as compared to the prior fiscal year due to an increase in deferred revenue. CIHR's deferred revenue balance fluctuates annually as it depends on the timing or receipt and disbursement of partner funds. CIHR's total departmental net debt as at March 31, 2014, did not materially change as compared to the prior fiscal year. Total non-financial assets declined by $2.1 million during the 2013-14 fiscal year due to the amortization of capital assets. The decline in net book value of CIHR's non-financial assets also led to the change in CIHR's departmental net financial position indicated in the table above.
Financial Statements
CIHR's 2013-14 audited financial statements can be found on the CIHR websiteFootnote 86 and form an integral part of the annual report. Included with this year's audited financial statements are the:
- Financial Statement Discussion and AnalysisFootnote 87;
- Auditor's Report and Financial StatementsFootnote 88 (including the Statement of Management Responsibility Including Internal Control over Financial Reporting); and
- Annex: Summary of the Assessment of Effectiveness of the Systems of Internal Control over Financial Reporting and the Action Plan of the Canadian Institutes of Health Research for the Fiscal Year 2012-13 (Unaudited).Footnote 89
Supplementary Information Tables
The supplementary information tables listed in the 2013-14 Departmental Performance Report can be found on the CIHR websiteFootnote 90.
- Departmental Sustainable Development Strategy
- Details on Transfer Payment Programs
- Internal Audits and Evaluations
Tax Expenditures and Evaluations
The tax system can be used to achieve public policy objectives through the application of special measures such as low tax rates, exemptions, deductions, deferrals and credits. The Department of Finance Canada publishes cost estimates and projections for these measures annually in the Tax Expenditures and EvaluationsFootnote 91 publication. The tax measures presented in the Tax Expenditures and Evaluations publication are the sole responsibility of the Minister of Finance.
Section IV: Organizational Contact Information
Canadian Institutes of Health Research
160 Elgin Street, 9th Floor
Address Locator 4809A
Ottawa, Ontario K1A 0W9
Canada
Telephone: 613-941-2672
Fax: 613-954-1800
Appendix: Definitions
- Appropriation:
- Any authority of Parliament to pay money out of the Consolidated Revenue Fund.
- Budgetary expenditures:
- Include operating and capital expenditures; transfer payments to other levels of government, organizations or individuals; and payments to Crown corporations.
- Departmental Performance Report:
- Reports on an appropriated organization's actual accomplishments against the plans, priorities and expected results set out in the corresponding Reports on Plans and Priorities. These reports are tabled in Parliament in the fall.
- Full-time equivalent:
- Is a measure of the extent to which an employee represents a full person-year charge against a departmental budget. Full-time equivalents are calculated as a ratio of assigned hours of work to scheduled hours of work. Scheduled hours of work are set out in collective agreements.
- Government of Canada outcomes:
- A set of 16 high-level objectives defined for the government as a whole, grouped in four spending areas: economic affairs, social affairs, international affairs and government affairs.
- Management, Resources and Results Structure:
- A comprehensive framework that consists of an organization's inventory of programs, resources, results, performance indicators and governance information. Programs and results are depicted in their hierarchical relationship to each other and to the Strategic Outcome(s) to which they contribute. The Management, Resources and Results Structure is developed from the Program Alignment Architecture.
- Non-budgetary expenditures:
- Include net outlays and receipts related to loans, investments and advances, which change the composition of the financial assets of the Government of Canada.
- Performance:
- What an organization did with its resources to achieve its results, how well those results compare to what the organization intended to achieve and how well lessons learned have been identified.
- Performance indicator:
- A qualitative or quantitative means of measuring an output or outcome, with the intention of gauging the performance of an organization, program, policy or initiative respecting expected results.
- Performance reporting:
- The process of communicating evidence-based performance information. Performance reporting supports decision making, accountability and transparency.
- Planned spending:
- For Reports on Plans and Priorities (RPPs) and Departmental Performance Reports (DPRs), planned spending refers to those amounts that receive Treasury Board approval by February 1. Therefore, planned spending may include amounts incremental to planned expenditures presented in the Main Estimates.
A department is expected to be aware of the authorities that it has sought and received. The determination of planned spending is a departmental responsibility, and departments must be able to defend the expenditure and accrual numbers presented in their RPPs and DPRs. - Plans:
- The articulation of strategic choices, which provides information on how an organization intends to achieve its priorities and associated results. Generally a plan will explain the logic behind the strategies chosen and tend to focus on actions that lead up to the expected result.
- Priorities:
- Plans or projects that an organization has chosen to focus and report on during the planning period. Priorities represent the things that are most important or what must be done first to support the achievement of the desired Strategic Outcome(s).
- Program:
- A group of related resource inputs and activities that are managed to meet specific needs and to achieve intended results and that are treated as a budgetary unit.
- Results:
- An external consequence attributed, in part, to an organization, policy, program or initiative. Results are not within the control of a single organization, policy, program or initiative; instead they are within the area of the organization's influence.
- Program Alignment Architecture:
- A structured inventory of an organization's programs depicting the hierarchical relationship between programs and the Strategic Outcome(s) to which they contribute.
- Report on Plans and Priorities:
- Provides information on the plans and expected performance of appropriated organizations over a three-year period. These reports are tabled in Parliament each spring.
- Strategic Outcome:
- A long-term and enduring benefit to Canadians that is linked to the organization's mandate, vision and core functions.
- Sunset program:
- A time-limited program that does not have an ongoing funding and policy authority. When the program is set to expire, a decision must be made whether to continue the program. In the case of a renewal, the decision specifies the scope, funding level and duration.
- Target:
- A measurable performance or success level that an organization, program or initiative plans to achieve within a specified time period. Targets can be either quantitative or qualitative.
- Whole-of-government framework:
- Maps the financial contributions of federal organizations receiving appropriations by aligning their Programs to a set of 16 government-wide, high-level outcome areas, grouped under four spending areas.
Endnotes
- Footnote 1
- Footnote 2
- Footnote 3
- Footnote 4
-
Minister of Health
- Footnote 5
- Footnote 6
- Footnote 7
- Footnote 8
- Footnote 9
- Footnote 10
- Footnote 11
-
Health Research Roadmap: Creating innovative research for better health and health care
- Footnote 12
- Footnote 13
- Footnote 14
-
Canada Graduate Scholarships
- Footnote 15
- Footnote 16
- Footnote 17
- Footnote 18
- Footnote 19
-
Science to Business
- Footnote 20
-
Science Policy Fellowships
- Footnote 21
-
Strategic Training Initiative in Health Research
- Footnote 22
-
Open Suite of Programs and peer review
- Footnote 23
-
Fellowship pilot
- Footnote 24
- Footnote 25
- Footnote 26
- Footnote 27
-
European Research Projects on Rare Diseases
- Footnote 28
- Footnote 29
- Footnote 30
- Footnote 31
- Footnote 32
- Footnote 33
-
Pilots held in 2013
- Footnote 34
-
External Advisory Committee Report: Training and Career Development in Patient-Oriented Research
- Footnote 35
-
SPOR National Steering Committee
- Footnote 36
- Footnote 37
-
The Reforms of the Open Suite of Programs
- Footnote 38
- Footnote 39
-
Designing for the Future: The New Open Suite of Programs and Peer Review Process
- Footnote 40
-
CIHR's Open Suite of Programs and peer review reforms
- Footnote 41
-
What CIHR Heard: Analysis of Feedback on the Design Discussion Document
- Footnote 42
- Footnote 43
-
Whole-of-government framework
- Footnote 44
- Footnote 45
- Footnote 46
- Footnote 47
- Footnote 48
- Footnote 49
- Footnote 50
-
Clinical Trials Summit Action Plan
- Footnote 51
- Footnote 52
-
Dr. Ilias Cagiannos
- Footnote 53
- Footnote 54
-
Dr. Igor Jurisica
- Footnote 55
-
Tools and resources
- Footnote 56
- Footnote 57
-
Evaluation of the Collaborative Health Research Projects [ PDF (552 KB) - external link ]
- Footnote 58
- Footnote 59
- Footnote 60
-
PC-TRIADD
- Footnote 61
- Footnote 62
- Footnote 63
-
Evaluation of the Regenerative Medicine and Nanomedicine Initiative
- Footnote 64
- Footnote 65
-
International review
- Footnote 66
-
Evaluation of the CIHR Clinical Trials Network in HIV/AIDS program
- Footnote 67
- Footnote 68
- Footnote 69
- Footnote 70
- Footnote 71
-
Pandemic Preparedness Strategic Research Initiative
- Footnote 72
-
Dr. Benedikt Fischer
- Footnote 73
- Footnote 74
-
Dr. Thomas Brown
- Footnote 75
- Footnote 76
-
Transformational Research in Adolescent Mental Health
- Footnote 77
-
CIHR and Graham Boeckh Foundation launch mental health research network (News Release)
- Footnote 78
-
SPOR capacity development
- Footnote 79
-
Patient engagement workshop
- Footnote 80
- Footnote 81
- Footnote 82
- Footnote 83
-
A Guide to Knowledge Synthesis
- Footnote 84
- Footnote 85
- Footnote 86
-
Canadian Institutes of Health Research Annual Report 2012–13: Innovative, Strategic, Current
- Footnote 87
-
Financial Statement Discussion and Analysis
- Footnote 88
-
Auditor's Report and Financial Statements
- Footnote 89
- Footnote 90
- Footnote 91
- Date modified: