2012-13 Departmental Performance Report
Table of Contents
- Minister's Message
- Section I: Organizational Overview
- Section II: Analysis of Programs and Sub-Programs by Strategic Outcome
- Section III: Supplementary Information
- Section IV: Other Items of Interest
- Endnotes
Minister's Message
 As Minister of Health, I am pleased to present the 2012-2013 Canadian Institutes of Health Research's (CIHR) Departmental Performance Report.
As Minister of Health, I am pleased to present the 2012-2013 Canadian Institutes of Health Research's (CIHR) Departmental Performance Report.
Our Government understands that a strong and vibrant research community plays a key role in improving the healthcare system and generating new economic opportunities. In the past 13 years, CIHR has steadily increased the impact of Canada's health research, demonstrating tangible benefits to Canadians, which has enhanced our country's reputation as an international leader in health science.
This year, CIHR released a design plan for a renewed Open Operating Grant Program and peer review process. Developed in close consultation with Canada's health research communities, these reforms are intended to ensure robust support for health research well into the future. Whether researchers are searching for the key to an HIV vaccine, or studying the fascinating world of the human microbiome, their work provides important information about the fundamentals of human health. We need to support researchers at all stages of their careers to pursue these questions.
CIHR drives innovation through investment in priority-driven research which mobilizes the research community and its partners. Health research and innovation make advances in healthcare possible and are very important to ensure the long-term sustainability of our healthcare system. That is why our Government has invested approximately $1 billion in CIHR to support Canada's health researchers and innovations and is funding over 14,000 health researchers across Canada.
CIHR leverages the strengths of its multiple Institutes and private and not-for-profit partners to build on Canada's research strengths. Our Government launched CIHR's Strategy for Patient-Oriented Research (SPOR), which is designed to ensure that patients receive the right treatment, at the right time, by putting research in the hands of healthcare providers. SPOR will focus on health challenges identified as priorities in multiple provinces and territories and pursue research to help bridge the gap between research evidence and healthcare practice.
As CIHR continues to explore new approaches for identifying and supporting excellent health research, it strengthens Canada's healthcare system and improves the lives of all Canadians.
The Honourable Rona Ambrose, PC, MP
Minister of Health
Section I: Organizational Overview
Raison d'être
CIHR1 is the Government of Canada's health research funding agency. It was created with a mandate2 to excel, according to internationally accepted standards of scientific excellence, in the creation of new knowledge and its translation into improved health for Canadians, more effective health services and products and a strengthened Canadian health care system.
Responsibilities
CIHR was designed to respond to the evolving needs for health research and seeks to transform health research in Canada by:
- funding both investigator-initiated research as well as research on targeted priority areas;
- building research capacity in underdeveloped areas and training the next generation of health researchers; and
- focusing on knowledge translation that facilitates the application of the results of research and their transformation into new policies, practices, procedures, products and services.
CIHR integrates research through a unique interdisciplinary structure made up of 13 "virtual" institutes3. These institutes are not "bricks-and-mortar" buildings but communities of experts and stakeholders. Each Institute supports a broad spectrum of research in its topic area: biomedical; clinical; health systems and services; and the social, cultural and environmental factors that affect the health of populations. Institutes form national research networks linking researchers, funders and knowledge users across Canada to work on priority areas.
CIHR reports through the Minister of Health4 and plays a key role in the Health Portfolio. As Canada's health research funding agency, CIHR makes an essential contribution to the Minister of Health's overall responsibilities by funding the research and knowledge translation needed to inform the evolution of Canadian health policy and regulation, and by taking an advisory role on research and innovation issues. This is achieved through an extensive and growing set of linkages with Health Canada5 and the Public Health Agency of Canada6, providing decision makers with access to high-quality and timely health research.
CIHR works closely with the Natural Sciences and Engineering Research Council7 (NSERC) and the Social Sciences and Humanities Research Council8 (SSHRC), the two granting councils of the Industry Portfolio, to share information and coordinate efforts, harmonize practices, avoid duplication and foster multidisciplinary research. The three organizations (referred to as "tri-agency") provide a channel for the implementation of common policies, practices and approaches, whenever possible.
CIHR's Governing Council9 (GC) sets the strategic direction of the agency and is responsible for evaluating its performance. Leadership on research, knowledge translation and funding for research is provided by the Science Council10 (SC), while leadership on corporate policy and management is provided by the Executive Management Committee11 (EMC).
Strategic Outcome(s) and Program Alignment Architecture
| Program 1.1: Health Knowledge |
Program 1.2: Health Researchers |
Program 1.3: Health Research Commercialization |
Program 1.4: Health and Health Services Advances |
Program 1.5: Internal Services |
|---|---|---|---|---|
|
||||
| Sub-Program 1.1.1 Open Research Grant Program |
Sub-Program 1.2.1 Salary Support Program |
Sub-Program 1.3.1 Research Commercialization Program |
Sub-Program 1.4.1 Institute Strategic Initiatives |
|
| Sub-Sub-Program 1.2.1.1 Open Salary Support Programs | Sub-Sub-Program 1.4.1.1 Strategic Initiatives Program | |||
| Sub-Sub-Program 1.2.1.2 Canada Research Chairs Program | Sub-Sub-Program 1.4.1.2 Institute Support Grants Program | |||
| Sub-Sub-Program 1.2.1.3 Canada Excellence Research Chairs Program | Sub-Sub-Program 1.4.1.3 HIV/AIDS Research Initiative | |||
| Sub-Sub-Program 1.4.1.4 Pandemic Preparedness Strategic Research Initiative | ||||
| Sub-Sub-Program 1.4.1.5 National Anti-Drug Strategy Treatment Research Initiative | ||||
| Sub-Sub-Program 1.4.1.6 Strategy for Patient-Oriented Research Initiative | ||||
| Sub-Sub-Program 1.4.1.7 Drug Safety and Effectiveness Network Initiative | ||||
| Sub-Program 1.1.2 RCTii Program |
Sub-Program 1.2.2 Training Support Program |
Sub-Program 1.3.2 NCEiii Program |
Sub-Program 1.4.2 Knowledge Translation Programs |
|
| Sub-Sub-Program 1.2.2.1 Open Training Support Programs | Sub-Sub-Program 1.3.2.1 Networks of Centres of Excellence Program | |||
| Sub-Sub-Program 1.2.2.2 Sir Frederick Banting and Dr. Charles Best Canada Graduate Scholarships | Sub-Sub-Program 1.3.2.2 Business-Led Networks of Centres of Excellence Program | |||
| Sub-Sub-Program 1.2.2.3 Georges Philias Vanier Canada Graduate Scholarships Program | Sub-Sub-Program 1.3.2.3 Centres of Excellence for Commercialization and Research Program | |||
Organizational Priorities
In 2009, CIHR's GC approved CIHR's second strategic plan Health Research Roadmap: Creating innovative research for better health and health care12 (2009-2014). This strategic plan is the product of widespread consultations with members of the health research community, careful assessment of Canada's strengths and weaknesses, and ongoing deliberation about what CIHR would like to achieve by 2014. Roadmap sets out a vision comprised of four strategic directions that are aligned with CIHR's corporate, business and operational priorities.
In 2010, CIHR implemented a rolling, three-year planning and reporting process for the implementation of Roadmap. The CIHR Three-Year Implementation Plan and Progress Report13 highlights the activities to be delivered on the strategic directions outlined in Roadmap. The report is updated annually in order to continuously assess progress made on achieving CIHR's strategic goals and priorities. The following table is a summary of CIHR's progress against the commitments made in the 2012-13 Report on Plans and Priorities (RPP), which are directly linked to Roadmap.
Summary of Progress Against Priorities
| Priority | Type | Programs |
|---|---|---|
|
||
| Invest in World-Class Research Excellence | Ongoing | 1.1 Health Knowledge 1.2 Health Researchers |
| Priority | Type | Program |
|---|---|---|
|
||
| Address Health and Health System Research Priorities | Ongoing | 1.4 Health and Health Services Advances |
| Priority | Type | Programs |
|---|---|---|
|
||
| Accelerate the Capture of Health and Economic Benefits of Health Research | Ongoing | 1.3 Health Research Commercialization 1.4 Health and Health Services Advances |
| Priority | Type | Program |
|---|---|---|
|
||
| Achieve Organizational Excellence, Foster Ethics and Demonstrate Impact | Ongoing | 1.5 Internal Services |
Risk Analysis
CIHR proactively identified, assessed and mitigated its corporate risks under the terms of the approved Corporate Risk Management Framework (Framework). The risk management governance structure contained in the Framework clearly defines the roles and responsibilities of risk owners, CIHR management, the Chief Risk Officer (CRO), Governing Council, and Audit Committee. The Framework also includes CIHR's Risk Management Policy, and describes the process and reporting requirements that have been established to develop and refresh CIHR's Corporate Risk Profile (CRP).
On a continuous basis, CIHR monitors and assesses both identified and potential risks. Throughout the year all risk owners are required to provide CIHR's CRO with updates to their risk mitigation strategies in order to ensure their overall strategy and implementation target dates are reasonable and meet the needs of the organization.
In order to satisfy the governance and accountability requirements of the Framework, both Governing Council and the Audit Committee receive regular reports on the issues relating to risk management, as well as information on any material changes to the Corporate Risk Profile from the CRO.
In 2012-13 CIHR identified 16 risks, of which five were considered high risks requiring mitigation and monitoring. These five risks are outlined below.
2012-13 Risk Table
| Risk 1 - Health Research Roadmap Implementation | Link to Program Alignment Architecture | Link to Organizational Priorities |
|---|---|---|
|
Risk Response Strategy: The Health Research Roadmap implementation risk was identified in the 2012-13 RPP and 2012-13 CRP, and CIHR responded to this risk by:
While progress has been made on the approved mitigation strategies, the impact, if the risk is not properly addressed, could result in loss of credibility from both key external and internal stakeholders and the public at large. As a result, the Health Research Roadmap implementation continues to be a high risk and is actively managed by CIHR on an ongoing basis. |
||
| In 2012-13 a risk was identified that CIHR may be unable to fully deliver on the reforms being made to the Open Suite of Programs, including changes to the peer review process. These changes are pivotal for the Health Research Roadmap Implementation. This includes the risk that external stakeholders do not understand or support the proposed changes to either the programs and/or peer review processes. | Strategic Outcome - A world-class health research enterprise that creates, disseminates and applies new knowledge across all areas of health research | Invest in World-Class Research Excellence |
| Risk 2 - Knowledge Translation | Link to Program Alignment Architecture | Link to Organizational Priorities |
|---|---|---|
|
Risk Response Strategy: The Knowledge Translation risk was identified in the 2012-13 RPP and 2012-13 CRP. CIHR responded to the risk by:
Knowledge Translation remains a high risk for CIHR and a renewed risk mitigation strategy has been developed. |
||
| Given CIHR's lack of direct control over factors influencing the uptake and use of the research it funds, in 2012-13 a risk was identified that CIHR may not be able to fully achieve the knowledge translation (KT) component of its mandate and improve the health of Canadians through health research. | Program 1.3 - Health Research Commercialization Program 1.4 - Health and Health Services Advances |
Accelerate the Capture of Health and Economic Benefits of Health Research |
| Risk 3 - Results Management and Monitoring | Link to Program Alignment Architecture | Link to Organizational Priorities |
|---|---|---|
|
Risk Response Strategy: The Results Management and Monitoring risk was identified in the 2012-13 RPP and 2012-13 CRP. CIHR responded to the risk by:
Results Management and Monitoring remains a high risk for CIHR. An updated mitigation strategy has been developed for 2013-14 and it is anticipated that once this strategy is implemented the risk will be reduced to medium. |
||
| Performance reporting and evaluation are time-consuming, costly and at times burdensome to target audiences. In 2012-13, a risk was identified that CIHR may be unable to optimally and efficiently evaluate and report on its performance as well as the results of funded research, which will compromise our ability to be accountable to Canadians. | Program 1.5 Internal Services | Achieve Organizational Excellence, Foster Ethics and Demonstrate Impact |
| Risk 4 - Institute Organizational Model | Link to Program Alignment Architecture | Link to Organizational Priorities |
|---|---|---|
|
Risk Response Strategy: The Institute Organizational Model risk was identified in the 2012-13 RPP and 2012-13 CRP. CIHR responded to this risk by:
The Institute Organizational Model risk could result in disruption to an Institute's business during the transition period (approximately 2 years); and not achieving the planned outcomes that each of the Institutes SDs commit to in their annual plan which could be critical for CIHR's business operations and strategic objectives. |
||
| Due to the Institute virtual organizational model, in 2012-13 CIHR identified a risk of disruptions and corporate memory loss during the transition period from the outgoing Scientific Director (SD) to the incoming. This may compromise the Institutes' abilities to achieve planned outcomes or their mandate in support of CIHR's strategic objectives. | Program1.4 Health and Health Services Advances Program 1.5 Internal Services |
Address Health and Health System Research Priorities Achieve Organizational Excellence, Foster Ethics and Demonstrate Impact |
| Risk 5 - Budgeting | Link to Program Alignment Architecture | Link to Organizational Priorities |
|---|---|---|
|
Risk Response Strategy: The Budgeting risk was identified in 2012-13 RPP and 2012-13 CRP and CIHR responded to this risk by:
|
||
| Given the diversity of expectations of the research community and CIHR's current budget, there is the possibility that CIHR will not have the flexibility to fully address the needs of the health research community as well as Canadians. | Strategic Outcome - A world-class health research enterprise that creates, disseminates and applies new knowledge across all areas of health research | Address Health and Health System Research Priorities Achieve Organizational Excellence, Foster Ethics and Demonstrate Impact |
Summary of Performance
| Total Budgetary Expenditures (Main Estimates) 2012-13 |
Planned Spending 2012-13 |
Total Authorities (available for use) 2012-13 |
Actual Spending (authorities used) 2012-13 |
Differenceiv (Planned vs. Actual Spending) |
|---|---|---|---|---|
|
||||
| 977.9 | 977.9 | 1,008.1 | 997.1 | 19.2 |
| Planned 2012-13 |
Actual 2012-13 |
Differencev (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
|
||
| 418 | 404 | 14 |
Summary of Performance Tables
Performance Summary Table for Strategic Outcome and Programs ($ millions)
Strategic Outcome: A world-class health research enterprise that creates, disseminates and applies new knowledge across all areas of health research
| Program | Total Budgetary Expenditures (Main Estimates 2012-13) | Planned Spending | Total Authorities (available for use 2012-13) |
Actual Spending (authorities used) | Alignment of Programs to Government of Canada Outcomes39 | ||||
|---|---|---|---|---|---|---|---|---|---|
| 2012-13 | 2013-14 | 2014-15 | 2012-13 | 2011-12 | 2010-11 | ||||
| 1.1 Health Knowledge | 451.6 | 451.0 | 473.3 | 473.2 | 452.4 | 483.0 | 470.5 | 468.5 | Healthy Canadians40 |
| 1.2 Health Researchers | 194.1 | 195.0 | 172.9 | 171.2 | 185.3 | 173.1 | 182.8 | 195.7 | |
| 1.3 Health Research Commercialization | 43.3 | 42.0 | 53.6 | 45.4 | 64.7 | 52.3 | 56.0 | 53.1 | |
| 1.4 Health and Health Services Advances | 261.1 | 260.9 | 253.8 | 252.6 | 276.0 | 260.9 | 268.0 | 275.4 | |
| Strategic Outcome Sub-Total | 950.1 | 948.9 | 953.6 | 942.4 | 978.4 | 969.3 | 977.3 | 992.7 | |
Performance Summary Table for Internal Services ($ millions)
| Internal Services | Total Budgetary Expenditures (Main Estimates 2012-13) | Planned Spending | Total Authorities (available for use 2012-13) |
Actual Spending (authorities used) | ||||
|---|---|---|---|---|---|---|---|---|
| 2012-13 | 2013-14 | 2014-15 | 2012-13 | 2011-12 | 2010-11 | |||
| 27.8 | 29.0 | 27.8 | 26.4 | 29.7 | 27.8 | 31.8 | 34.2 | |
| Internal Services Sub-Total | 27.8 | 29.0 | 27.8 | 26.4 | 29.7 | 27.8 | 31.8 | 34.2 |
Total Performance Summary Table ($ millions)
| Strategic Outcome and Internal Services | Total Budgetary Expenditures (Main Estimates 2012-13) | Planned Spending | Total Authorities (available for use 2012-13) |
Actual Spending (authorities used) | ||||
|---|---|---|---|---|---|---|---|---|
| 2012-13 | 2013-14 | 2014-15 | 2012-13 | 2011-12 | 2010-11 | |||
| 977.9 | 977.9 | 981.4 | 968.8 | 1,008.1 | 997.1 | 1,009.1 | 1,026.9 | |
| Total | 977.9 | 977.9 | 981.4 | 968.8 | 1,008.1 | 997.1 | 1,009.1 | 1,026.9 |
Spending and FTE Variance Explanations
CIHR's Total Authorities of $1,008.1M in 2012-13, have seen an increase of $30.2M compared to its planned spending. The increase in Total Authorities is primarily the result of funding received through Budget 2012. This includes $15.0M for CIHR's Strategy for Patient-Oriented Research. CIHR also received additional funding for the fourth round of grants of the Centres of Excellence for Commercialization and Research ($14.3M), and the Business-Led Networks of Centres of Excellence grants received $7.8M.
There was a net increase of approximately $3.0M in transfers from or to other government departments for key initiatives such as Aboriginal health, influenza research and population health interventions. The remaining $4.5M increase was the result of a $2.1M operating budget carry forward from the previous year and $2.4M in technical adjustments to cover employee severance, salaries and other related benefits and entitlements.
These increases in Total Authorities are offset by a $14.4M decrease as a result of the streamlining of operations and the implementation of improved efficiencies. The savings measures implemented included reductions to CIHR's tri-agency programs ($9.1M), the wind-up of the Regenerative Medicine and Nanomedicine initiatives ($3.1M), and the streamlining of CIHR's operations ($2.2M).
In 2012-13, CIHR's actual expenditures totalled $997.1M, which was $11.0M less than its Total Authorities. CIHR has requested Treasury Board approval to reprofile $7.7M of these unspent funds to future fiscal years for the Centres of Excellence for Commercialization and Research and the Business-Led Networks of Centres of Excellence grants. CIHR is also carrying forward $2.4M in unspent authorities from its operating expenditure vote to the 2013-14 fiscal year.
Planned FTEs were higher than actual FTEs due to the streamlining of operations and the implementation of efficiency measures.
Expenditure Profile
Departmental Spending Trend
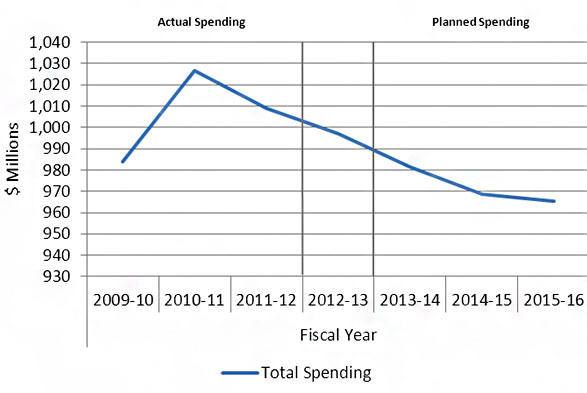
| 2009-10 | 2010-11 | 2011-12 | 2012-13 | 2013-14 | 2014-15 | 2015-16 | |
|---|---|---|---|---|---|---|---|
| Total Spending | 983.8 | 1,026.9 | 1,009.1 | 997.1 | 981.4 | 968.8 | 965.5 |
CIHR's actual spending increased from $983.8M in 2009-10 to reach $1,026.9M in 2010-11, and then decreased to $1,009.1M in 2011-12. Actual spending for 2012-13 was $997.1M. In the absence of new funding, total planned spending will decrease annually in 2013-14, 2014-15, and 2015-16 as indicated in the graph above.
The variations in departmental spending from $983.8M in 2009-10 to $1,009.1M in 2011-12 are mainly due to several permanent increases to CIHR's budget announced in Federal Budgets over this period, coupled with incremental funding for targeted programs such as the Canada Excellence Research Chairs ($7.7M), the Drug Safety and Effectiveness Network (DSEN) ($5.1M), the Vanier Canada Graduate Scholarships ($5.1M) and the Banting Postdoctoral Fellowships ($1.6M).
Other reasons for the variance include variations in annual funding for two of CIHR's tri-agency programs, the Centres of Excellence for Commercialization and Research (CECR) ($14.3M) and the Business-Led Networks of Centres of Excellence (BL-NCE) ($7.8M). CIHR's share of these tri-agency programs' funding is not fixed but rather determined by the results of program competitions. Therefore, the decrease in planned spending in future fiscal years could be offset by additional authorities granted for upcoming CECR and BL-NCE competitions.
Estimates by Vote
For information on CIHR's organizational Votes and/or statutory expenditures, please see Public Accounts of Canada 2013 (Volume II).41 An electronic version of the Public Accounts 2013 is available on the Public Works and Government Services Canada website.42
Strategic Environmental Assessment
During 2012-13, CIHR considered the environmental effects of initiatives subject to the Cabinet Directive on the Environmental Assessment of Policy, Plan and Program Proposals43 and did not produce any public statements.
Section II: Analysis of Programs and Sub-Programs by Strategic Outcome
Strategic Outcome: A world-class health research enterprise that creates, disseminates and applies new knowledge across all areas of health research.
CIHR supports health research in order to improve the health of Canadians and to deliver more effective health care services to Canadians.
CIHR uses peer review processes to identify exemplary projects and individuals that merit funding. In 2012-13, approximately 3000 peer reviewers provided their time, without remuneration, and served on 238 peer review committee meetings to help review over 12,919 applications. Without the voluntary support from this community of experts, CIHR would not have the necessary financial and human resources needed to review the applications with the intellectual rigor needed to ensure relevancy and scientific excellence.
Program 1.1: Health Knowledge
Program Description:
This program aims to support the creation of new knowledge across all areas of health research to improve health and the health system. This is achieved by managing CIHR's open competition and related peer review processes based on internationally accepted standards of scientific excellence.
2012-13 Financial Resources and Human Resources (FTEs)
| Total Budgetary Expenditures (Main Estimates) 2012-13 |
Planned Spending 2012-13 |
Total Authorities (available for use) 2012-13 | Actual Spending (authorities used) 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|---|---|
| 451.6 | 451.0 | 452.4 | 483.0 | 32.0 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 81 | 109 | 28 |
| Program Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
| Health research advances knowledge. | Outputs and impacts of CIHR-funded research. | Maintain or increase | CIHR met the 2012-13 target by increasing expenditures to $468.7M, which is an increase over the 2011-12 result of $459.7M. For details concerning the impacts of this result, please see the Investing in Optogenetics Research box as well as the Performance Analysis and Lessons Learned Section for Sub-Program 1.1.1 Open Research Grant Program. |
Performance Analysis and Lessons Learned
The variance between planned spending and actual spending of $32.0M is largely due to the high number of quality applications received via the Open Operating Grant Program (OOGP), as well as an in-year reallocation from Program 1.2 to the OOGP. The variance between planned and actual FTEs is related to an internal reorganization at CIHR during 2012-13, a number of FTE's were re-assigned to better align with the organization's shifting priorities and the correlating decrease of staff can be seen in Program 1.2 for example.
CIHR provided funding to support research in all areas of health and achieved its target of providing 800 new multi-year grants through two open competitions in March and September. In 2012-13, CIHR funded a total of 803 new multi-year grants for a total committed value of $ 479.8M. CIHR received and reviewed a total of 4,617 applications through these two grant competitions.
CIHR invested $468.7M in the Open Operating Grant Program44 in 2012-13, as compared to $459.7M in 2011-12, an increase of 1.9%. In 2012-13, CIHR supported a total of 3,967 multi-year grants through the OOGP as compared to 3,946 in 2011-12. The average annual grant payment increased from $116,507 in 2011-12 to $118,148 in 2012-13.
In an effort to optimize the peer review system within the OOGP, CIHR developed a triage and streamlining approach which was piloted during the spring 2012 OOGP competition. By triaging a set proportion of the applications based on their initial scores, committee reviewers were able to spend less time on applications found to have little chance of being funded and more time discussing grants that required full discussion (normally those applications that fell within the grey zone, which are a cohort of applications that typically fall between the funding cut-off and the ceiling). By spending more time on these applications, committee reviewers were able to improve the efficiency with which the final rankings within the committee are determined. This new approach is now employed throughout CIHR's open programs.
Sub-Program 1.1.1 Open Research Grant Program
Program Description:
The Open Research Grant Program provides operating funds to support research proposals in all areas of health research. The Open Operating Grant Program is the largest component of this program. Competitions are typically held each March and September with an open call for research proposals, with no restrictions on areas of research or maximum level of requested funds.
2012-13 Financial Resources – Sub-Program Level and Human Resources (FTEs)
| Planned Spending 2012-13 |
Actual Spending 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|
| 420.6 | 470.4 | 49.8 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 76 | 106 | 30 |
2012-13 Performance Results
| Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
|
|||
| Health research is conducted and translated. | Number of publications from CIHR-supported research | Maintain or increase | In 2012-13 CIHR-supported research publications totalled 5,751 which is an unexpectedly large increase over the 2011-12 result of 2,858. CIHR believes the main reason for this apparent increase in publications is an improvement to the tracking methodology resulting from the implementation of our Research Reporting System. |
| Knowledge Translation activities of funded researchers | Maintain or increase | In 2012-13 there were 22,069vi KT activities. This is a significant increase over the 2011-12 results of 6,444. CIHR believes the main reason for this apparent increase in KT activities is an improvement to the tracking methodology resulting from the implementation of our Research Reporting System. | |
Performance Analysis and Lessons Learned
For variance explanations regarding spending and FTEs please see Program 1.1 Health Knowledge.
All open and strategic programs at CIHR require end of grant reporting. In 2012-13, the Research Reporting System, which is an end-of-grant reporting module collecting information from CIHR-funded researchers on the results of their grant(s), received close to 1,100 reports which were mainly from grants in CIHR's Open Operating Grant Program. Data from RRS45 for grants with expiry dates between April 1st, 2009 and March 31, 2013 received in 2012-13 stated that these grants produced 5,751 journal publications. The data from RRS for the same period also indicated that 22,069 KT activities by researchers occurred, including 10,756 presentations and 5,562 interviews.
Researchers supported by CIHR thought the OOGP have made significant contributions to the advancement of knowledge. For example, in an annual report, ScienceWatch46, a web resource for science analysis run by Thomson Reuters47, monitors researchers according to citations during 2012. This report has declared Dr. Salim Yusuf's ,who has worked on 19 CIHR grants, to be one of the hottest researchers48 based on the number of highly cited papers (Dr. Yusuf has 11 highly cited papers). Another example, captured by CIHR's RRS in 2012-13, is Dr. R.A. Hegele, a researcher who has been active for almost three decades, recently acknowledged the contribution CIHR has made towards supporting his research. Dr. Hegele has an H-index49 of 52 (meaning that 52 of his publications have at least 52 citations each, as per Scopus 16-07-201350). He has been receiving continuous CIHR funding since 1998 and believes that his publication and citation record provides the strongest validation of the lab's research. He reported that the research results of his CIHR-supported project have resulted in several national and international research awards, produced 58 original articles and 27 review articles since 2004. He also reported in his 2012-13 RRS report that as of December 31, 2011, these articles have been cited 1,163 times demonstrating knowledge translation resulting from CIHR funding.
Evaluations were completed for two components of the Sub-Program Open Research Grant Program. This included a summative evaluation of the Interagency Advisory Panel and Secretariat on Research Ethics51 which was completed in 2009 and an evaluation of the Open Operating Grant Program52 completed in 2012.
Sub-Program 1.1.2 Randomized Controlled Trials Program
Program Description:
The Randomized Controlled Trials Program supports experiments to evaluate the efficiency and effectiveness of interventions in health or health services by randomly assigning individuals to receive or not receive one or more interventions that are being compared. The results are analyzed by comparing outcomes in the different groups.
2012-13 Financial Resources – Sub-Program Level and Human Resources (FTEs)
| Planned Spending 2012-13 |
Actual Spending 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|
| 30.4 | 12.6 | 17.8 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 5 | 3 | 2 |
2012-13 Performance Results
| Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
| High-quality evidence on the efficacy and effectiveness of interventions in health and health services | Evidence produced by funded trials | All completed trials have demonstrated whether or not interventions under study are effective or not | Not available. As of June 2009, CIHR's Randomized Controlled Trials Program has been integrated into the Open Research Grant Program and therefore, cannot be reported separately. |
Performance Analysis and Lessons Learned
The variances for actual spending and FTEs related to Sub-Program 1.1.2 Randomized Controlled Trials (RCT) are due to the winding down of the remaining grant commitments from previous years as the program integrates with the Open Research Grant Program. The correlating increase in spending and FTEs can be found in Sub-Program 1.1.1 Open Research Grant Program and Program 1.1 Health Knowledge.
An example of the impact of CIHR funding is the establishment of the Ontario Best Practices Research Initiative 53 (OBRI). The OBRI is a collaboration of rheumatology stakeholders representing rheumatologists, patients, researchers and others. The mission of the OBRI is to provide the optimal use of treatments for the management of Ontarians living with Rheumatoid Arthritis (RA). CIHR funded the initial protocol which was designed as a prospective controlled study comparing the real world effectiveness of biologics to traditional disease-modifying antirheumatic drugs in adults with RA residing in Ontario. Today OBRI has 58 rheumatologists recruiting patients and 2289 patients have been referred. Those who consent to participate in OBRI are now followed for a minimum of five years. The overall goal of the OBRI was originally limited to a pharmacology platform to assess the long-term effectiveness, sustainability and safety of pharmacologic treatments for RA in actual practice (with expansion to other types of inflammatory arthritis (IA)). The evolution of the OBRI research platform has now been extended to inform policy and clinical decision making through clinical practice monitoring and other interventions targeted to health system efficiencies.
Program 1.2: Health Researchers
Program Description:
This program aims to build health research capacity to improve health and the health system by supporting the training and careers of excellent health researchers through a competitive peer review process based on internationally accepted standards of scientific excellence.
2012-13 Financial Resources and Human Resources (FTEs)
| Total Budgetary Expenditures (Main Estimates) 2012-13 |
Planned Spending 2012-13 |
Total Authorities (available for use) 2012-13 | Actual Spending (authorities used) 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|---|---|
| 194.1 | 195.0 | 185.3 | 173.1 | 21.9 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 33 | 15 | 18 |
2012-13 Performance Results
| Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
|
|||
| A strong and talented health research community with the capacity to undertake health research. | Number of graduate trainees in Canada compared to international levelsvii. | Maintain or increase international ranking. | CIHR met the 2012-13 target by increasing the number of PhD graduates in Canada per 100,000 aged 25-29 increased from 191 graduates per 100,000 in 2005 to 226 in 2010. Canada is ranked 6th out of the G7 for PhD graduates in per 100,000 population aged 25-29. |
| Number and fields of investigators and trainees funded. | Maintain number and diversity (by theme and Institute domain) of trainees funded. | CIHR closely met the 2012-13 target by funding 2,538 investigators and trainees in all areas of research in all themesviii (as opposed to 2,797 in 2011-12). | |
Performance Analysis and Lessons Learned
The variance between planned spending and actual spending of $21.9M is related to the Canada Research Chairs (CRC) program and the streamlining of operations and the implementation of improved efficiencies, as well as an in-year reallocation of unspent authorities from the CRC program to the Open Operating Grant Program (under Program 1.1). The variance between planned and actual FTEs is related to an internal reorganization at CIHR during 2012-13, a number of FTEs were reassigned to other programs to better align with the organization's shifting priorities.
CIHR provides funding to support health researchers throughout their graduate training and beyond. Through various competitions in 2012-13, CIHR provided funding for 762 new awards (training and salary combined). These awards represent a total commitment of $138.4M which includes expenditures of $31.2M by CIHR in 2012-13 to support trainees and health researchers.
Additionally, over 1,300 trainees are currently being supported through the CIHR Strategic Training Initiative in Health Research (STIHR) 54, which was launched in 2008. An evaluation of STIHR 55 was completed in 2008.
In 2012-13, CIHR continued to support diversity by funding excellence in all areas of its mandate (by theme and Institute-specific research area). The total number of investigators supported by CIHR decreased from 2,797 in 2011-12 to 2,538 in 2012-13. The relative drop of researchers and trainees supported by CIHR in 2012-13 can be attributed to the winding down of temporary funding resulting from Canada's Economic Action Plan (EAP) 56, which provided an additional 400 Canada Graduate Scholarship (CGS) 57 master's scholarships in 2009 and 2010 competitions. EAP also provided an additional 200 doctoral scholarships each year from 2009 to 2012.
Sub-Program 1.2.1 Salary Support Program
Program Description:
The Salary Support Program provides salary support to help new health researchers develop their careers and devote more time to initiating and conducting health research.
2012-13 Financial Resources – Sub-Program Level and Human Resources (FTEs)
| Planned Spending 2012-13 |
Actual Spending 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|
| 125.6 | 111.5 | 14.1 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 21 | 10 | 11 |
2012-13 Performance Results
| Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
| Excellent health researchers available to undertake health research | Types and number of health researchers funded | Maintain or increase | CIHR met the 2012-13 target by supporting the following number of new health researchers:
|
| Number of fundable applicants who are funded | Maintain or increase | CIHR nearly met the 2012-13 target by funding 921 health researchers which is a decrease compared to the 969 health researchers supported in 2011-12. The relative decrease of researchers and trainees supported by CIHR in 2012-13 is attributable to the temporary funding from Canada's Economic Action Plan (EAP) coming to an end. |
Performance Analysis and Lessons Learned
For explanations on spending and FTEs please see Program 1.2 Health Researchers.
In 2012-13, CIHR invested through the Salary Support Program $109.2M by funding 921 health researchers, in an effort to build research capacity to improve health and the health system. A total of 130 new awards were provided, representing a total commitment of $85.3M over the next 8 years which includes expenditures of $10.8M in 2012-13 to support health researchers.
CIHR through the Open Salary Support Programs, in 2012-13 funded a total of 38 new investigators which provided these outstanding new investigators with the opportunity to develop and demonstrate their independence in initiating and conducting health research through provision of a contribution to their salary. These new awards represent a total commitment of $11.8M over the next 6 years, which includes expenditures of $1.5M in 2012-13.
Furthermore, CIHR funded 92 new awards through the Canada Research Chairs Program (CRC)58; this represents a total commitment of $73.5M over the next 8 years, including expenditures of $9.4M in 2012-13 to attract and retain some of the world's most accomplished and promising researchers. CIHR provides two levels of Awards: Tier 1 Chairs are for outstanding researchers acknowledged by their peers as world leaders in their fields, and Tier 2 Chairs are for exceptional emerging researchers, acknowledged by their peers as having the potential to lead in their field. CIHR awarded a total of 32 new Tier 1 Chairs (for $5.1M) and 60 new Tier 2 Chairs (for $4.3M) in 2012-13.
Evaluations completed for this Sub-Program include: the Salary/Career Award Programs59 in 2012 and the Canada Research Chairs program60 in 2010.
Sub-Program 1.2.2 Training Support Program
Program Description:
The Training Support Program provides support and special recognition to PhD, post-PhD or post health professional degree students who are training in health research areas in Canada or abroad.
2012-13 Financial Resources – Sub-Program Level and Human Resources (FTEs)
| Planned Spending 2012-13 |
Actual Spending 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|
| 69.4 | 61.6 | 7.8 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 12 | 5 | 7 |
2012-13 Performance Results
| Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
| Highly trained individuals available for health research | Numbers of trainees completing degrees or fellowship programs | Maintain or increase | CIHR mostly met the target of developing capacity of highly trained researchers in health research by supporting 1,467 trainees. Access to data on degree completion is limited. CIHR implemented an end-of-grant reporting system which will allow for future reporting on this indicator. |
Performance Analysis and Lessons Learned
For explanations on spending and FTEs please see Program 1.2 Health Researchers.
In 2012-13, CIHR through the Training Support Program invested $60.7M by funding 1,467 trainees including master's, doctoral, postdoctoral or (post) health professional degrees in Canada and abroad. Through these competitions, 632 new awards were offered which represent a total commitment of $53.0M over the next 6 years and includes expenditures of $20.3M in 2012-13 to support highly trained individuals.
The Vanier Canada Graduate Scholarships program was launched in fiscal year 2008-09 and was created as a tri-agency program, in partnership with SSHRC and NSERC. The purpose of this program is to strengthen Canada's ability to attract and retain the world's top-tier doctoral students. In the 2012-13 competition, CIHR funded 53 new Vanier Canada Graduate Scholarship61 awards, 13 of these awards were given to foreign citizens. These recipients are world-class doctoral students who demonstrate a high standard of scholarly achievement in graduate studies as well as leadership skills.
In 2012-13, CIHR also funded 23 new Banting Postdoctoral fellowships62 awards 5 of which were awarded to foreign citizens. These recipients are all top-tier national and international postdoctoral fellows. The Banting fellowships will enable them to develop their leadership potential, will position them for success as research leaders of tomorrow, and will contribute to Canada's economic, social and research-based growth through support of research-intensive careers.
In 2011-12, the presidents of Canada's three federal granting agencies approved the creation of a joint Vanier Banting Secretariat. Since its inception, the Vanier Banting Secretariat has greatly improved communication channels and increased program delivery efficiencies through the harmonization of processes, guidelines and policies which came to effect in 2012-13.
Finally, CIHR invested $9.3M through the Canada Graduate Scholarships (CGS)63 program in order to support the learning and development of its health research trainees by awarding 174 new doctoral awards and 180 new master's awards.
An evaluation of the Canada Graduate Scholarships64 program was completed in 2009.
Program 1.3: Health Research Commercialization
Program Description:
This program aims to support and facilitate the commercialization of health research to improve health and the health system. This is achieved by managing funding competitions to provide grants, in partnership with the private sector where relevant, using peer review processes based on internationally accepted standards of scientific excellence, and by building and strengthening the capacity of Canadian health researchers to engage in the commercialization process.
2012-13 Financial Resources and Human Resources (FTEs)
| Total Budgetary Expenditures (Main Estimates) 2012-13 |
Planned Spending 2012-13 |
Total Authorities (available for use) 2012-13 | Actual Spending (authorities used) 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|---|---|
| 43.3 | 42.0 | 64.7 | 52.3 | 10.3 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 7 | 7 | 0 |
2012-13 Performance Results
| Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
| Commercial activity – products (patents and intellectual property), companies and employment generated. | Outputs and impacts of funded research. | Evidence that some work of CIHR funded researchers resulted in some of these long-term impacts | CIHR mostly met the 2012-13 target with expenditures totalling $52.3 million. This is a slight decrease from the 2011-12 spending of $55.3M. For details concerning the impacts of this result, please see the Performance Analysis and Lessons Learned below. |
Performance Analysis and Lessons Learned
Planned spending was $10.3M less than actual spending, primarily due to the fact that throughout the year CIHR received additional funding of $22.1M to fund the fourth round of grants for the Centres of Excellence for Commercialization and Research (CECR) and for the Business-Led Networks of Centres of Excellence (BL-NCE) grants. This $22.1M increase in funding received was offset by a request to reprofile $7.7M in funds to 2013-14. The impact on funding was minimalized by a further decrease of $1.6M related to the BL-NCE program.
In 2012-13, CIHR spent a total of $52.3M which represents 181 grants in Health Research Commercialization programs, including 100 new grants funded totalling $68.4M in new commitments and expenditures of $26.4M. In 2011-12 CIHR spent a total of $55.3M in Health Research Commercialization. Therefore expenditures decreased by 5.8% for 2012-13.
CIHR also took over the administration of the joint CIHR/NSERC program, the Collaborative Health Research Projects (CHRP). The NSERC-partnered CHRP program supports innovative interdisciplinary collaborative research projects with a strong KT component and requiring participation from the natural sciences or engineering community together with the health sciences community. In 2012-13, CIHR funded 37 CHRP new grants for a total commitment of $10.9M.
CIHR launched Phase 1 and Phase 2 of the Proof of Principle (POP) competition in fall 2012 which resulted in 31 new grants and a total commitment of $5.1M in funding.
CIHR-funded research has had a positive impact on the commercialization of new health products. For example a new pharmaceutical developed by CIHR-funded researcher Dr. Daniel Drucker of Mount Sinai Hospital's Lunenfeld-Tanenbaum Research Institute65 has been approved by the U.S. Food and Drug Administration to treat patients with short bowel syndrome. The drug, called teduglutide or Gattex®, is marketed by NPS Pharmaceuticals and represents the first advance in a long-term treatment option for the debilitating condition in almost 40 years. Short bowel syndrome occurs in patients who have had half or more of their small intestine removed – usually due to Crohn's disease, trauma, ischemic bowel injury or bowel cancer. As a result, patients cannot absorb enough water, vitamins and nutrients and must rely on intravenous feeding for provision of nutrients and to maintain hydration. The new drug promotes repair and normal growth of the intestinal lining, decreases energy loss and increases nutrient absorption. In follow-up long-term clinical studies, one in seven patients treated with teduglutide were able to discontinue intravenous nutrition. Dr. Drucker is internationally renowned for his work in the field of gut hormones and their role in diabetes, obesity and inflammatory bowel disorders.
Sub-Program 1.3.1 Research Commercialization Program
Program Description:
The Research Commercialization Program is comprised of a suite of funding initiatives that aim to support the creation of new knowledge, practices, products and services and to facilitate the commercialization of this knowledge.
2012-13 Financial Resources – Sub-Program Level and Human Resources (FTEs)
| Planned Spending 2012-13 |
Actual Spending 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|
| 17.0 | 13.9 | 3.1 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 3 | 3 | 0 |
2012-13 Performance Results
| Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
| Health research is commercialized more effectively | Outputs and impacts of funded research | Maintain or increase | CIHR achieved over 80% of its target in 2012-13, with expenditures totalling $13.9 million. This is a decrease from the 2011-12 spending of $16.8 million. For details concerning the impacts of this sub-program, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
For explanations on spending variances please see Program 1.3 Health Research Commercialization.
In 2012-13, CIHR spent a total of $13.9M in Research Commercialization programs, which represents 164 grants. CIHR's Proof of Principle Program aims to facilitate and improve the commercial transfer of knowledge and technology resulting from academic health research for the benefit of Canadians.
Other reports from program managers and other sources show that CIHR-funded research has had a positive impact on the commercialization of new health products. An example is a potential cancer treatment using a virus to kill cancer cells. Dr. Patrick Lee is using a naturally occurring, benign virus that does not cause any major illnesses in humans and is proving effective against cancer cells. Since 1998, when he identified the reovirus as a cancer killer in mice, Dalhousie University 66 virologist Dr. Patrick Lee has steadily expanded the understanding of how the virus infects a cancer cell. The reovirus replicates within a cancer cell to produce thousands of particles that eventually rupture it. The released virus particles then seek out neighboring cancer cells to repeat the process, leaving normal cells untouched. Dr. Lee's current work adds further evidence that the virus also works to stimulate the body's immune system to attack tumours. Oncolytics Biotech Inc. of Calgary, whose Chief Operating Officer, Dr. Matt Coffey, co-authored the original 1998 paper with Dr. Lee, is working to bring the reovirus to the clinic with a therapy called Reolysin®. The company is conducting a 14-country Phase III clinical trial – the last step before securing approval to take a new treatment to market – using intravenously administered Reolysin in combination with chemotherapy to kill head and neck tumours.
An evaluation of the College and Community Innovation Program67 under this Sub-Program was completed in 2013.
Sub-Program 1.3.2 Networks of Centres of Excellence (NCE) Program
Program Description:
The Networks of Centres of Excellence Program supports the networking of centres of research excellence with industrial know-how and strategic investment to turn Canadian research and entrepreneurial talent into economic and social benefits for Canada. The Networks of Centres of Excellence Program is national in scope, multidisciplinary and involve multisectoral partnerships.
2012-13 Financial Resources – Sub-Program Level and Human Resources (FTEs)
| Planned Spending 2012-13 |
Actual Spending 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|
| 25.0 | 38.4 | 13.4 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 4 | 4 | 0 |
2012-13 Performance Results
| Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
| World-class networks and centres connect leading-edge research with industrial know-how and strategic investment | Significant demonstration of impacts | 100% of networks demonstrate impacts by year 4 | CIHR met this target given the fact that all networks demonstrated significant impact. For details concerning the impacts of this sub-program, please see the Performance Analysis and Lessons Learned section below. |
| Ratio of partner contributions to NCE funding by networks at year 4 | Greater than or equal to 1 | CIHR exceeded this target with a ratio of partner contribution greater than 1. |
Performance Analysis and Lessons Learned
For explanations on spending variances please see Program 1.3 Health Research Commercialization.
CIHR's NCE Sub-Program includes the Networks of Centres of Excellence (NCE); Centres of Excellence for Commercialization and Research (CECR)68 and the Business-Led Networks of Centres of Excellence program69 (BL-NCE). CIHR administered $38.4M in grants and awards during 2012-13 which is equivalent with 2011-12. For NCE specifically, CIHR funded 17 grants and awards in 2012-13.
Reports from program managers and other sources show that partnerships fostered by the tri-agency Networks of Centres of Excellence (NCE) program meets its objectives. The 2011-12 NCE report70 (which is the most recently published report) demonstrated that the NCE had a significant impact with 267 patent applications filed; 145 new products and 87 licences granted or under negotiation. Overall, tri-agency investment in the NCE demonstrated an increased capacity to develop, to test and to accelerate promising technologies to new products and innovations.
The 2011-12 NCE report demonstrates that the NCE, CECR and BL-NCE are very effective tools for stimulating private sector investment in research innovation. Overall, the ratio of partner contributions to NCE funding is $1.59 for every NCE dollar and exceeds the target of greater than or equal to 1.
Various evaluations of the activities contained in this Sub-Program have been completed over the past few years including: the Centres of Excellence for Commercialization and Research Program71 in 2012; the Business-Led Networks of Centres of Excellence program72 in 2012; and two evaluation reports in 2009 of the Networks of Centres of Excellence program's International Partnerships Initiative73 and New Initiatives74.
Program 1.4: Health and Health Services Advances
Program Description:
Through the competitive peer review process based on internationally accepted standards of scientific excellence, this program aims to support the creation of new knowledge in strategic priority areas and its translation into improved health and a strengthened health system.
2012-13 Financial Resources and Human Resources (FTEs)
| Total Budgetary Expenditures (Main Estimates) 2012-13 |
Planned Spending 2012-13 |
Total Authorities (available for use) 2012-13 | Actual Spending (authorities used) 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|---|---|
| 261.1 | 260.9 | 276.0 | 260.9 | 0.0 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 106 | 86 | 20 |
2012-13 Performance Results
| Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
| Translation and use of health research takes place | Impacts of CIHR funded research | Anecdotal evidence that work of CIHR funded researchers resulted in some long-term impact | For details concerning the anecdotal evidence of CIHR funded researchers of this program, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
Planned FTEs were higher than actual FTEs due to an internal reorganization which resulted in operations being streamlined and improved efficiencies being implemented.
CIHR has many funding mechanisms which were used in 2012-13 to facilitate all aspects of KT from knowledge synthesis to uptake. Below are examples from both signature and institute initiatives of how some of these tools have been successful in pushing knowledge from 'bench to bedside to best-practice'.
CIHR's Evidence-Informed Healthcare Renewal (EIHR)75 signature initiative aims to provide timely and high-quality evidence on how best to finance, sustain and govern provincial, territorial and federal health care systems. In 2012-13, 57 integrated knowledge translation (KT) projects were funded totaling $3.9M, aimed at addressing priority topics identified by health policy and decision makers. An EIHR Roundtable76, to facilitate health care renewal discussions and sharing of information among more than 30 organizations continued in 2012-13. Participating organizations created a "one-stop-shop" repository77 of health care renewal evidence, facilitated by CIHR. This repository is a good example of a strong network for sharing of health care renewal evidence. Furthermore, EIHR-funded researchers produced seven knowledge syntheses which are currently being disseminated to stakeholders through various KT means for maximum uptake and impact.
In June 2012, researchers funded by the Institute of Circulatory and Respiratory Health78 announced a web-based tool allowing physicians to estimate the risk of dying within seven days of presentation to hospital among acute heart failure patients. Use of this tool can reduce the admission rate for cardiovascular disease- Canada's leading cause of hospitalization.
Finally, as part of CIHR's Inflammation in Chronic Disease79 Signature Initiative, the Canadian National Transplant Research Program80 (CNTRP) was launched in 2013, uniting transplant researchers with critical care research communities in an effort to develop new knowledge and health care practices which will address barriers to tissue and organ donation, thus improving health outcomes for transplant recipients in Canada. To date, 105 researchers across 9 provinces have collaborated to address barriers to donation, increase the number and quality of available organs, and improve quality of life and long-term survival rates of patients. Funding is provided through CIHR, in partnership with various national and provincial services and foundations, as well as industry, hospital and academic partners.
CIHR recognizes the importance of identifying lessons learned and using the potentials of its experience by evaluating challenges of these programs or processes in order to take corrective actions or to develop future directions. CIHR put emphasis on creating a vision to demonstrate management's engagement of continuous improvement. For instance, by being open to new and innovative partnerships that CIHR could not have anticipated when the initiative started, CNTRP will result in a higher number of partners than originally anticipated. This vision allows CIHR to be more effective, efficient and receptive to unplanned, innovative partnership initiatives.
Sub-Program 1.4.1 Institute Strategic Initiatives
Program Description:
The Institute Strategic Initiatives program is led by CIHR's 13 Institutes and funds grants and awards to support research and researchers in priority areas to address strategic health opportunities, threats and challenges to Canadians. The Institutes identify these strategic priority research areas in consultation with stakeholders from government, health care, patient and community groups, and industry. Proposals are solicited from researchers by issuing a Request for Applications outlining the specific theme/area where research is needed. Applications are peer reviewed using criteria specific to the funding opportunity.
2012-13 Financial Resources – Sub-Program Level and Human Resources (FTEs)
| Planned Spending 2012-13 |
Actual Spending 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|
| 240.7 | 238.0 | 2.7 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 98 | 78 | 20 |
2012-13 Performance Results
| Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
| Health research is conducted, translated and used in priority areas | Outputs and impacts of funded research and success against objectives | Maintain or increase | CIHR met the 2012-13 target by funding 2,310 strategic initiative grants, which is an increase over the 2011-12 result of 2,260. For details concerning the impacts of this sub-program, please see the Performance Analysis and Lessons Learned section below. |
Performance Analysis and Lessons Learned
Planned FTEs were higher than actual FTEs due to an internal reorganization which resulted in operations being streamlined and improved efficiencies being implemented.
In an effort to achieve CIHR's goal of addressing health and health system research priorities, CIHR created signatures81 and strategic initiatives82 to attain greater focus and impact from our strategic investments. CIHR signature and strategic initiatives achieved the following in 2012-13:
- CIHR and partners funded 12 Community-Based Primary Health Care83 innovation teams.
- As part of the International Collaborative Research Strategy for Alzheimer's Disease84 signature initiative, CIHR launched the Canadian Consortium on Neurodegeneration in Aging85.
- CIHR announced the results of a major $150.0M partnership with Genome Canada86; part of the Personalized Medicine Signature Initiative87. Seventeen projects in total have been approved for funding, which will focus on the application of genomics to tailor patient treatments and therapies in fields as diverse as epilepsy, autism, HIV/AIDS, cancer, cardiovascular disease, rare neurological diseases, and stroke, among others.
- CIHR invested $23.5M in strategic HIV/AIDS research grants and capacity building programs through its HIV/AIDS Research Initiative88, supported by the Federal Initiative to Address HIV/AIDS in Canada and the Canadian HIV Vaccine Initiative.
- Drug Safety and Effectiveness Network89 (DSEN) research was taken up by decision makers90. For example, a study by the DSEN-funded Canadian Collaboration for Drug Safety, Effectiveness and Network Meta-Analysis informed a therapeutic review by the Canadian Agency for Drugs and Technologies in Health on the treatment of atrial fibrillation, contributing several key messages on the use of new oral anticoagulants91.
- CIHR invested in several research projects on Multiple Sclerosis (MS). As an example, CIHR led a national coalition supporting a Phase I/II clinical trial on Chronic Cerebrospinal Venous Insufficiency (CCSVI) in individuals living with MS. The clinical trial, which began on November 1, 2012, has a total budget of approximately $6.0M over four years. This study will determine whether the procedure is safe and efficient for MS patients.
- The Global Alliance for Chronic Disease92 (GACD), a consortium of medical and health research funders including CIHR, has agreed to fund 15 teams from 19 countries to tackle the global health issue of hypertension, for a total investment of $23.0M. GACD-funded teams came together in Ottawa for the first annual meeting of the joint technical steering committee in 2012.
- CIHR launched the Patient-Oriented Network in Adolescent and Youth Mental Health in October 2012. The network is the first launched under the Strategy for Patient-Oriented Research93, which aims to improve the health care system and health outcomes by providing the right clinical intervention to the right patient at the right time. The network is a $25.0M collaborative effort between CIHR and the Graham Boeckh Foundation94.
Sub-Program 1.4.2 Knowledge Translation Programs
Program Description:
The Knowledge Translation (KT) Programs consist of a suite of funding opportunities that aim to support the synthesis, dissemination, exchange and ethically sound application of knowledge in any area of health research. These programs support the science of KT, capacity development in KT science, and integrated KT- collaborative research which involves researchers and knowledge users working together to address relevant research questions and to exchange and apply knowledge to help solve health and health system problems.
2012-13 Financial Resources – Sub-Program Level and Human Resources (FTEs)
| Planned Spending 2012-13 |
Actual Spending 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|
| 20.2 | 22.9 | 2.7 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 8 | 8 | 0 |
2012-13 Performance Results
| Expected Results | Performance Indicators | Targets | Actual Results |
|---|---|---|---|
| Health research is translated more effectively | Number of knowledge translation activities supported by CIHR (and its partners where relevant) or resulting from CIHR activities (e.g. synthesis papers, briefs, participation in policy task forces, etc.) | Maintain or increase | CIHR met the 2012-13 target with an average of 16.9 KT activities per grant. |
| Number of stakeholders engaged in CIHR KT | Maintain or increase | As part of the KT programs, participation for stakeholders is required and therefore based on the evaluation outlined below CIHR engaged 373 stakeholders through 373 grants between 2005 and 2010. | |
| Outputs and impacts of funded research and success against objectives | Maintain or increase | CIHR mostly met the 2012-13 target by funding 538 knowledge translation grants, which is a decrease from the 2011-12 result of 671. The decrease is a result of the winding down of some of the funding opportunities which were transferred to the Open Operating Grant Program. |
Performance Analysis and Lessons Learned
In 2012-13, CIHR supported 538 Knowledge Translation grants and spent $21.9M on Knowledge Translation Programs as compared to $27.8M in 2011-12. The decrease from $27.8M in 2011-12 to $21.9M in 2012-13 is mostly attributable to 2011-12 representing the last year of three-year funding to pilot the Knowledge Translation Supplement Grant Program, which represented an investment of $5.9M in 2011-12. An evaluation of the knowledge translation programs began in 2012-13, and data collected to date shows that during the period from 2005 to 2010, an average of 16.9 KT activities per grant could be estimated from the 373 grants supported through three integrated knowledge translation programs (Partnerships for Health System Improvement95 (PHSI), Knowledge Synthesis, and Knowledge to Action).
The integrated knowledge translation programs require the participation of a knowledge user throughout the research process; these individuals are often decision makers who are in a position to influence policy or practice. This eligibility requirement lets us presume that a minimum of 373 stakeholders were engaged. In addition, the PHSI program requires a minimum partnership contribution (cash or in-kind). These design elements help leverage funds/resources and/or facilitate collaboration between researchers and individuals or organizations with the goal to move health research into policies, practices, products or services.
An evaluation was completed in 2012-13, and the final report will be published in 2013-14. The findings of this report will be used to ensure the strengths of the Knowledge Translation programs are captured in CIHR's new open suite program design.
Program 1.5: Internal Services
Program Description:
Internal Services are groups of related activities and resources that are administered to support the needs of programs and other corporate obligations of an organization. These groups are: Management and Oversight Services; Communications Services; Legal Services; Human Resources Management Services; Financial Management Services; Information Management Services; Information Technology Services; Real Property Services; Materiel Services; Acquisition Services; and Travel and Other Administrative Services. Internal Services include only those activities and resources that apply across an organization and not to those provided specifically to a program.
2012-13 Financial Resources and Human Resources (FTEs)
| Total Budgetary Expenditures (Main Estimates) 2012-13 |
Planned Spending 2012-13 |
Total Authorities (available for use) 2012-13 | Actual Spending (authorities used) 2012-13 |
Difference (Planned vs. Actual Spending) 2012-13 |
|---|---|---|---|---|
| 27.8 | 29.0 | 29.7 | 27.8 | 1.2 |
| Planned 2012-13 |
Actual 2012-13 |
Difference (Planned vs. Actual FTEs) 2012-13 |
|---|---|---|
| 191 | 187 | 4 |
Performance Analysis and Lessons Learned
Planned spending was slightly higher than actual spending primarily due to a $1.0M reduction in CIHR's operating budget as a result of the streamlining of operations and the implementation of improved efficiencies. CIHR also carried forward $2.4M of its operating budget to 2013-14. These decreases were offset by additional funding of $2.3M for technical adjustments to cover employee salaries and other related benefits. The remainder of the reduction is due to operating efficiencies and reduced costs for peer review.
CIHR was able to achieve significant savings with respect to its operating expenditures during 2012-13. In 2012-13, total operating expenditures decreased by approximately $2.1M (or 3.6%) in comparison to 2011-12 due to an internal reorganization at CIHR in which operations were streamlined and improved efficiencies were identified, thus reducing CIHR's salary and employee benefit expenditures. CIHR was also able to significantly reduce both hospitality expenditures (12.0%) and travel expenditures (3.4%) as compared to the 2011-12 fiscal year through its expanded use of virtual peer review meetings.
In order to address the changing focus of CIHR's work, recent budgetary reductions and increasing complexity of both external partnerships and funding opportunities, the Research and Knowledge Translation Portfolio at CIHR underwent a major reorganization. As a result of the reorganization, the Platforms and Major Initiatives (PMI) Branch was created with the major goal of increasing internal capacity to develop, manage and monitor large-scale strategic initiatives and enhance performance measurement tools and practices. This will allow CIHR to better monitor the progress of significant strategic investments and provide ongoing feedback to both grant recipients and CIHR governance bodies. Ultimately, it will contribute to CIHR's overarching goal to improve the health of Canadians and the efficiency and effectiveness of the health care system.
In 2012–13, CIHR's Evaluation Unit conducted a comprehensive evaluation which determined that the OOGP is succeeding in its primary role of supporting knowledge creation, reporting that researchers funded by the program produce publications with a greater scientific impact than the health research average for Canada. The program also plays a significant role in supporting capacity development in Canada's health research sector. The evaluation determined that an average of 8.61 research staff are trained on each open operating grant. Based on available data, the total number trained for all grants is estimated at 81,175 OOGP research staff between 2000 and 2010. The report has provided valuable and timely input into the current open suite program reform process for the OOGP.
Section III: Supplementary Information
Financial Statements Highlights
Condensed Statement of Operations and Departmental Net Financial Position
| 2012–13 Planned Results |
2012–13 Actual |
2011-12 Actual |
$ Change (2012-13 Planned and Actual) |
$ Change (2012-13 Actual vs. 2011-12 Actual) |
|
|---|---|---|---|---|---|
| Total expenses | 990.9 | 1,011.9 | 1,020.5 | (21.0) | (8.6) |
| Total revenues | 12.6 | 16.0 | 14.8 | (3.4) | 1.2 |
| Net cost of operations before government funding and transfers | 978.3 | 995.9 | 1,005.7 | (17.6) | (9.8) |
| Departmental net financial position | 1.6 | 0.3 | (0.5) | 1.3 | 0.8 |
Overall, total planned expenses are $21.0M lower than actual expenses incurred in 2012-13. This is primarily due to additional funds received throughout the year through the supplementary estimates. CIHR received an additional $15.0M through Budget 2012, as well as additional funding of $22.1M for the Centres of Excellence for Commercialization and Research and for the Business-Led Networks of Centres of Excellence grants. There was an aggregate increase of $3.0M due to various net transfers from or to other government departments. The remaining increase is due to a $2.1M operating budget carry forward from the previous year and $2.3M in technical adjustments to cover employee severance, salaries and other related benefits and entitlements. These increases were offset by a $14.4M net decrease to CIHR's 2012-13 budget as a result of the streamlining of operations and the implementation of improved efficiencies.
Overall, total actual expenses decreased by $8.6M in 2012-13 compared to 2011-12. This is primarily due to the EAP funding of $6.9M coming to an end related to the Canada Graduate Scholarships program (program continues), the Pandemic Research Initiative ($2.4M) not being renewed and increased expenses of $0.6M in the Vanier Canada Graduate Scholarships.
Actual total revenue was higher than the prior year actual and higher than planned as a result of more money being received than expected from collaborators during the fiscal year to fund programs.
Condensed Statement of Financial Position
| 2012-13 | 2011–12 | Change | |
|---|---|---|---|
| Total net liabilities | 12.1 | 15.1 | (3.0) |
| Total net financial assets | 9.3 | 10.8 | (1.5) |
| Departmental net debt | 2.8 | 4.3 | (1.5) |
| Total non-financial assets | 3.1 | 3.8 | (0.7) |
| Departmental net financial position | 0.3 | (0.5) | 0.8 |
Total net liabilities and net financial assets decreased by $3.0M and $1.5M, respectively, over the prior year, having a positive impact on the Departmental net debt. These corresponding decreases resulted directly from the following:
- CIHR's termination of employee severance provisions and payments made to employees during 2012-13, which resulted in a decreased liability of $1.5M;
- Recognition of additional revenues in 2012–13, whereby CIHR disbursed funds on behalf of external parties to fund additional health research grants and awards, which resulted in a decreased liability of $2.4M; and
- The decrease in assets is primarily a result of the overall reduction in liabilities noted above as well as the increased amortization of tangible capital assets in 2012–13.
Financial Statements
CIHR's 2012-13 Audited Financial Statements can be found on the CIHR website 96 and form an integral part of the Annual Report. Included with this year's audited financial statements are the:
- Statement of Management Responsibility Including Internal Control Over Financial Reporting;
- Financial Statements and notes; and
- Annex: Summary of the Assessment of Effectiveness of the Systems of Internal (with Action Plan).
List of Supplementary Information Tables
All electronic supplementary information tables listed in the 2012–13 Departmental Performance Report can be found on CIHR website97.
- Details on Transfer Payment Programs
- Greening Government Operations
- Internal Audits and Evaluations
- Sources of Respendable and Non-Respendable Revenue
Tax Expenditures and Evaluations Report
The tax system can be used to achieve public policy objectives through the application of special measures such as low tax rates, exemptions, deductions, deferrals and credits. The Department of Finance publishes cost estimates and projections for these measures annually in the Tax Expenditures and Evaluations publication98. The tax measures presented in the Tax Expenditures and Evaluations publication are the sole responsibility of the Minister of Finance.
Section IV: Other Items of Interest
Organizational Contact Information
Canadian Institutes of Health Research
160 Elgin Street, 9th Floor
Address Locator 4809A
Ottawa, Ontario
K1A 0W9
Email: support-soutien@cihr-irsc.gc.ca
Telephone: 613-954-1968
Toll Free: 1-888-603-4178
Fax: 613-954-1800
Endnotes
Footnotes
- Footnote 1
- Footnote 2
- Footnote 3
- Footnote 4
-
Minister of Health
- Footnote 5
- Footnote 6
- Footnote 7
- Footnote 8
- Footnote 9
- Footnote 10
- Footnote 11
- Footnote 12
-
Health Research Roadmap: Creating innovative research for better health and health care
- Footnote 13
-
CIHR Three-Year Implementation Plan and Progress Report
- Footnote 14
- Footnote 15
-
Open Operating Grant Program Competitions
- Footnote 16
- Footnote 17
- Footnote 18
-
CIHR's Canadian Epigenetics, Environment and Health Research Consortium Signature Initiative
- Footnote 19
- Footnote 20
-
Pathways to Health Equity for Aboriginal Peoples Signature Initiative
- Footnote 21
-
Expedited Knowledge Synthesis
- Footnote 22
- Footnote 23
-
Northwest Territories Department of Health and Social Services
- Footnote 24
- Footnote 25
- Footnote 26
- Footnote 27
- Footnote 28
- Footnote 29
- Footnote 30
- Footnote 31
- Footnote 32
- Footnote 33
- Footnote 34
-
2011 CIHR International Review Report
- Footnote 35
- Footnote 36
- Footnote 37
-
Policy to Access on Research Outputs
- Footnote 38
-
Research Reporting System
- Footnote 39
- Footnote 40
- Footnote 41
- Footnote 42
- Footnote 43
-
Cabinet Directive on the Environmental Assessment of Policy, Plan and Program Proposals
- Footnote 44
-
Open Operating Grant Program
- Footnote 45
-
Data from RRS
- Footnote 46
- Footnote 47
- Footnote 48
- Footnote 49
- Footnote 50
- Footnote 51
- Footnote 52
- Footnote 53
- Footnote 54
-
CIHR Strategic Training Initiative in Health Research (STIHR) Program
- Footnote 55
-
Evaluation of STIHR
- Footnote 56
-
Economic Action Plan (EAP)
- Footnote 57
- Footnote 58
- Footnote 59
- Footnote 60
-
Canada Research Chairs program [ PDF (1.68 MB) - external link ]
- Footnote 61
- Footnote 62
- Footnote 63
- Footnote 64
-
Canada Graduate Scholarships
- Footnote 65
-
Mount Sinai Hospital's Lunenfeld-Tanenbaum Research Institute
- Footnote 66
- Footnote 67
-
College and Community Innovation Program [ PDF (1.00 MB) - external link ]
- Footnote 68
-
Centres of Excellence for Commercialization and Research (CECR)
- Footnote 69
- Footnote 70
- Footnote 71
-
Centres of Excellence for Commercialization and Research Program [ PDF (887 KB) - external link ]
- Footnote 72
- Footnote 73
-
International Partnerships Initiative [ PDF (127 KB) - external link ]
- Footnote 74
- Footnote 75
- Footnote 76
- Footnote 77
- Footnote 78
- Footnote 79
- Footnote 80
- Footnote 81
- Footnote 82
- Footnote 83
- Footnote 84
-
International Collaborative Research Strategy for Alzheimer's Disease
- Footnote 85
- Footnote 86
- Footnote 87
- Footnote 88
- Footnote 89
- Footnote 90
-
Taken up by decision makers
- Footnote 91
-
For more information, see the supplementary information table of Health Canada's 2012-13 DPR about the horizontal initiative called Food and Consumer Safety Action Plan
- Footnote 92
- Footnote 93
- Footnote 94
- Footnote 95
- Footnote 96
- Footnote 97
- Footnote 98
- Date modified: